Label: DEXLIDO KIT- dexamethasone sodium phosphate, lidocaine hydrochloride, povidine iodine kit
- NDC Code(s): 55150-158-72, 63323-506-01, 67777-419-02, 76420-767-01
- Packager: Asclemed USA, Inc.
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated October 5, 2024
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- Dexamethasone Sodium Phosphate Injection, USP
-
DESCRIPTION
Dexamethasone Sodium Phosphate Injection, USP, is a water-soluble inorganic ester of dexamethasone which produces a rapid response even when injected intramuscularly.
Dexamethasone Sodium Phosphate, USP chemically is Pregna-1,4-diene-3,20-dione, 9-fluoro- 11,17-dihydroxy-16-methyl-21-(phosphonooxy)-, disodium salt, (11ß, 16α).
It occurs as a white to creamy white powder, is exceedingly hygroscopic, is soluble in water and its solutions have a pH between 7.0 and 8.5. It has the following structural formula:
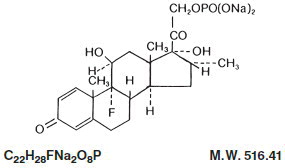
Each mL of Dexamethasone Sodium Phosphate Injection, USP (Preservative Free) contains dexamethasone sodium phosphate, USP equivalent to 10 mg dexamethasone phosphate; 24.75 mg sodium citrate, dihydrate; and Water for Injection, q.s. pH adjusted with citric acid or sodium hydroxide, if necessary. pH: 7.0 to 8.5.
-
CLINICAL PHARMACOLOGY
Dexamethasone sodium phosphate injection has a rapid onset but short duration of action when compared with less soluble preparations. Because of this, it is suitable for the treatment of acute disorders responsive to adrenocortical steroid therapy.
Naturally occurring glucocorticoids (hydrocortisone and cortisone), which also have salt-retaining properties, are used as replacement therapy in adrenocortical deficiency states. Their synthetic analogs, including dexamethasone, are primarily used for their potent anti-inflammatory effects in disorders of many organ systems.
Glucocorticoids cause profound and varied metabolic effects. In addition, they modify the body’s immune responses to diverse stimuli.
At equipotent anti-inflammatory doses, dexamethasone almost completely lacks the sodium-retaining property of hydrocortisone and closely related derivatives of hydrocortisone.
-
INDICATIONS AND USAGE
By intravenous or intramuscular injection when oral therapy is not feasible:
1. Endocrine Disorders
Primary or secondary adrenocortical insufficiency (hydrocortisone or cortisone is the drug of choice; synthetic analogs may be used in conjunction with mineralocorticoids
where applicable; in infancy, mineralocorticoid supplementation is of particular importance).
Acute adrenocortical insufficiency (hydrocortisone or cortisone is the drug of choice; mineralocorticoid supplementation may be necessary, particularly when synthetic analogs
are used).
Preoperatively, and in the event of serious trauma or illness, in patients with known adrenal insufficiency or when adrenocortical reserve is doubtful.
Shock unresponsive to conventional therapy if adrenocortical insufficiency exists or is suspected.
Congenital adrenal hyperplasia
Nonsuppurative thyroiditis
Hypercalcemia associated with cancer
2. Rheumatic Disorders
As adjunctive therapy for short-term administration (to tide the patient over an acute episode or exacerbation) in:
Post-traumatic osteoarthritis
Synovitis of osteoarthritis
Rheumatoid arthritis, including juvenile rheumatoid arthritis (selected cases may require low-dose maintenance therapy).
Acute and subacute bursitis
Epicondylitis
Acute nonspecific tenosynovitis
Acute gouty arthritis
Psoriatic arthritis
Ankylosing spondylitis
3. Collagen Diseases
During an exacerbation or as maintenance therapy in selected cases of:
Systemic lupus erythematosus
Acute rheumatic carditis
4. Dermatologic Diseases
Pemphigus
Severe erythema multiforme (Stevens-Johnson syndrome)
Exfoliative dermatitis
Bullous dermatitis herpetiformis
Severe seborrheic dermatitis
Severe psoriasis
Mycosis fungoides
5. Allergic States
Control of severe or incapacitating allergic conditions intractable to adequate trials of conventional treatment in:
Bronchial asthma
Contact dermatitis
Atopic dermatitis
Serum sickness
Seasonal or perennial allergic rhinitis
Drug hypersensitivity reactions
Urticarial transfusion reactions
Acute noninfectious laryngeal edema (epinephrine is the drug of first choice).
6. Ophthalmic Diseases
Severe acute and chronic allergic and inflammatory processes involving the eye, such as:
Herpes zoster ophthalmicus
Iritis, iridocyclitis
Chorioretinitis
Diffuse posterior uveitis and choroiditis
Optic neuritis
Sympathetic ophthalmia
Anterior segment inflammation
Allergic conjunctivitis
Keratitis
Allergic corneal marginal ulcers
7. Gastrointestinal Diseases
To tide the patient over a critical period of the disease in:
Ulcerative colitis (systemic therapy)
Regional enteritis (systemic therapy)
8. Respiratory Diseases
Symptomatic sarcoidosis
Berylliosis
Fulminating or disseminated pulmonary tuberculosis when used concurrently with appropriate antituberculous chemotherapy.
Loeffler’s syndrome not manageable by other means.
Aspiration pneumonitis
9. Hematologic Disorders
Acquired (autoimmune) hemolytic anemia.
Idiopathic thrombocytopenic purpura in adults
(IV only; IM administration is contraindicated).
Secondary thrombocytopenia in adults
Erythroblastopenia (RBC anemia)
Congenital (erythroid) hypoplastic anemia
10. Neoplastic Diseases
For palliative management of:
Leukemias and lymphomas in adults
Acute leukemia of childhood
11. Edematous States
To induce diuresis or remission of proteinuria in the nephrotic syndrome, without uremia, of the idiopathic type or that due to lupus erythematosus.
12. Miscellaneous
Tuberculous meningitis with subarachnoid block or impending block when used concurrently with appropriate antituberculous chemotherapy.
Trichinosis with neurologic or myocardial involvement.
13. Diagnostic testing of adrenocortical hyperfunction.
14. Cerebral Edema associated with primary or metastatic brain tumor, craniotomy, or head injury. Use in cerebral edema is not a substitute for careful neurosurgical evaluation
and definitive management such as neurosurgery or other specific therapy.
- CONTRAINDICATIONS
-
WARNINGS
Because rare instances of anaphylactoid reactions have occurred in patients receiving parenteral corticosteroid therapy, appropriate precautionary measures should be taken prior to administration, especially when the patient has a history of allergy to any drug. Anaphylactoid and hypersensitivity reactions have been reported for dexamethasone sodium phosphate injection (see ADVERSE REACTIONS).
Corticosteroids may exacerbate systemic fungal infections and, therefore, should not be used in the presence of such infections unless they are needed to control drug reactions due to amphotericin B. Moreover, there have been cases reported in which concomitant use of amphotericin B and hydrocortisone was followed by cardiac enlargement and congestive failure.
In patients on corticosteroid therapy subjected to any unusual stress, increased dosage of rapidly acting corticosteroids before, during, and after the stressful situation is indicated.
Drug-induced secondary adrenocortical insufficiency may result from too rapid withdrawal of corticosteroids and may be minimized by gradual reduction of dosage. This type of relative insufficiency may persist for months after discontinuation of therapy; therefore, in any situation of stress occurring during that period, hormone therapy should be reinstituted. If the patient is receiving steroids already, dosage may have to be increased. Since mineralocorticoid secretion may be impaired, salt and/or a mineralocorticoid should be administered concurrently.
Corticosteroids may mask some signs of infection, and new infections may appear during their use. There may be decreased resistance and inability to localize infection when corticosteroids are used. Moreover, corticosteroids may affect the nitroblue-tetrazolium test for bacterial infection and produce false negative results.
In cerebral malaria, a double-blind trial has shown that the use of corticosteroids is associated with prolongation of coma and a higher incidence of pneumonia and gastrointestinal bleeding.
Corticosteroids may activate latent amebiasis. Therefore, it is recommended that latent or active amebiasis be ruled out before initiating corticosteroid therapy in any patient who has spent time in the tropics or in any patient with unexplained diarrhea.
Prolonged use of corticosteroids may produce posterior subcapsular cataracts, glaucoma with possible damage to the optic nerves, and may enhance the establishment of secondary ocular infections due to fungi or viruses.
Average and large doses of cortisone or hydrocortisone can cause elevation of blood pressure, salt and water retention, and increased excretion of potassium. These effects are less likely to occur with the synthetic derivatives except when used in large doses. Dietary salt restriction and potassium supplementation may be necessary. All corticosteroids increase calcium excretion.
Administration of live virus vaccines, including smallpox, is contraindicated in individuals receiving immunosuppressive doses of corticosteroids. If inactivated viral or bacterial vaccines are administered to individuals receiving immunosuppressive doses of corticosteroids, the expected serum antibody response may not be obtained. However, immunization procedures may be undertaken in patients who are receiving corticosteroids as replacement therapy, e.g., for Addison’s disease.
Patients who are on drugs which suppress the immune system are more susceptible to infections than healthy individuals. Chickenpox and measles, for example, can have a more serious or even fatal course in non-immune children or adults on corticosteroids. In such children or adults who have not had these diseases, particular care should be taken to avoid exposure. The risk of developing a disseminated infection varies among individuals and can be related to the dose, route and duration of corticosteroid administration as well as to the underlying disease. If exposed to chickenpox, prophylaxis with varicella zoster immune globulin (VZIG) may be indicated. If chickenpox develops, treatment with antiviral agents may be considered. If exposed to measles, prophylaxis with immune globulin (IG) may be indicated. (See the respective package inserts for VZIG and IG for complete prescribing information).
The use of dexamethasone sodium phosphate injection in active tuberculosis should be restricted to those cases of fulminating or disseminated tuberculosis in which the corticosteroid is used for the management of the disease in conjunction with an appropriate antituberculous regimen.
If corticosteroids are indicated in patients with latent tuberculosis or tuberculin reactivity, close observation is necessary as reactivation of the disease may occur. During prolonged corticosteroid therapy, these patients should receive chemoprophylaxis.
Literature reports suggest an apparent association between use of corticosteroids and left ventricular free wall rupture after a recent myocardial infarction; therefore, therapy with corticosteroids should be used with great caution in these patients.
Serious Neurologic Adverse Reactions with Epidural Administration
Serious neurologic events, some resulting in death, have been reported with epidural injection of corticosteroids. Specific events reported include, but are not limited to, spinal cord infarction, paraplegia, quadriplegia, cortical blindness, and stroke. These serious neurologic events have been reported with and without use of fluoroscopy. The safety and effectiveness of epidural administration of corticosteroids has not been established, and corticosteroids are not approved for this use.
Usage in Pregnancy
Since adequate human reproduction studies have not been done with corticosteroids, use of these drugs in pregnancy or in women of childbearing potential requires that the anticipated benefits be weighed against the possible hazards to the mother and embryo or fetus. Infants born of mothers who have received substantial doses of corticosteroids during pregnancy should be carefully observed for signs of hypoadrenalism.
Corticosteroids appear in breast milk and could suppress growth, interfere with endogenous corticosteroid production, or cause other unwanted effects. Mothers taking pharmacologic doses of corticosteroids should be advised not to nurse.
-
PRECAUTIONS
This product, like many other steroid formulations, is sensitive to heat. Therefore, it should not be autoclaved when it is desirable to sterilize the exterior of the vial.
Following prolonged therapy, withdrawal of corticosteroids may result in symptoms of the corticosteroid withdrawal syndrome including fever, myalgia, arthralgia, and malaise. This may occur in patients even without evidence of adrenal insufficiency.
There is an enhanced effect of corticosteroids in patients with hypothyroidism and in those with cirrhosis.
Corticosteroids should be used cautiously in patients with ocular herpes simplex for fear of corneal perforation.
The lowest possible dose of corticosteroid should be used to control the condition under treatment, and when reduction in dosage is possible, the reduction must be gradual.
Psychic derangements may appear when corticosteroids are used, ranging from euphoria, insomnia, mood swings, personality changes, and severe depression to frank psychotic manifestations. Also, existing emotional instability or psychotic tendencies may be aggravated by corticosteroids.
Aspirin should be used within caution in conjunction with corticosteroids in hypoprothrombinemia.
Steroids should be used with caution in nonspecific ulcerative colitis, if there is a probability of impending perforation, abscess, or other pyogenic infection, also in diverticulitis, fresh intestinal anastomoses, active or latent peptic ulcer, renal insufficiency, hypertension, osteoporosis, and myasthenia gravis. Signs of peritoneal irritation following gastrointestinal perforation in patients receiving large doses of corticosteroids may be minimal or absent. Fat embolism has been reported as a possible complication of hypercortisonism.
When large doses are given, some authorities advise that antacids be administered between meals to help prevent peptic ulcer.
Steroids may increase or decrease motility and number of spermatozoa in some patients.
Phenytoin, phenobarbital, ephedrine, and rifampin may enhance the metabolic clearance of corticosteroids resulting in decreased blood levels and lessened physiologic activity, thus requiring adjustment in corticosteroid dosage. These interactions may interfere with dexamethasone suppression tests which should be interpreted with caution during administration of these drugs.
False negative results in the dexamethasone suppression test (DST) in patients being treated with indomethacin have been reported. Thus, results of the DST should be interpreted with caution in these patients.
The prothrombin time should be checked frequently in patients who are receiving corticosteroids and coumarin anticoagulants at the same time because of reports that corticosteroids have altered the response to these anticoagulants. Studies have shown that the usual effect produced by adding corticosteroids is inhibition of response to coumarins, although there have been some conflicting reports of potentiation not substantiated by studies.
When corticosteroids are administered concomitantly with potassium-depleting diuretics, patients should be observed closely for development of hypokalemia.
The slower rate of absorption by intramuscular administration should be recognized.
-
ADVERSE REACTIONS
Fluid and electrolyte disturbances:
Sodium retention
Fluid retention
Congestive heart failure in susceptible patients
Potassium loss
Hypokalemic alkalosis
Hypertension
Musculoskeletal:
Muscle weakness
Steroid myopathy
Loss of muscle mass
Osteoporosis
Vertebral compression fractures
Aseptic necrosis of femoral and humeral heads
Tendon rupture
Pathologic fracture of long bones
Gastrointestinal:
Peptic ulcer with possible subsequent perforation and hemorrhage
Perforation of the small and large bowel; particularly in patients with inflammatory
bowel disease
Pancreatitis
Abdominal distention
Ulcerative esophagitis
Dermatologic:
Impaired wound healing
Thin fragile skin
Petechiae and ecchymoses
Erythema
Increased sweating
May suppress reactions to skin tests
Burning or tingling, especially in the perineal area (after IV injection)
Other cutaneous reactions, such as allergic dermatitis, urticaria, angioneurotic edema
Neurologic:
Convulsions
Increased intracranial pressure with papilledema (pseudotumor cerebri) usually after
treatment
Vertigo
Headache
Psychic disturbances
Endocrine:
Menstrual irregularities
Development of cushingoid state
Suppression of growth in pediatric patients
Secondary adrenocortical and pituitary unresponsiveness, particularly in times of
stress, as in trauma, surgery, or illness
Decreased carbohydrate tolerance
Manifestations of latent diabetes mellitus
Increased requirements for insulin or oral hypoglycemic agents in diabetics
Hirsutism
Ophthalmic:
Posterior subcapsular cataracts
Increased intraocular pressure
Glaucoma
Exophthalmos
Retinopathy of prematurity
Metabolic:
Negative nitrogen balance due to protein catabolism
Cardiovascular:
Myocardial rupture following recent myocardial infarction (see WARNINGS)
Hypertrophic cardiomyopathy in low birth weight infants
Other:
Anaphylactoid or hypersensitivity reactions
Thromboembolism
Weight gain
Increased appetite
Nausea
Malaise
Hiccups
The following additional adverse reactions are related to parenteral corticosteroid therapy:
Hyperpigmentation or hypopigmentation
Subcutaneous and cutaneous atrophy
Sterile abscess
Charcot-like arthropathy
-
OVERDOSAGE
Reports of acute toxicity and/or death following overdosage of glucocorticoids are rare. In the event of overdosage, no specific antidote is available; treatment is supportive and symptomatic.
The oral LD 50 of dexamethasone in female mice was 6.5 g/kg. The intravenous LD 50 of dexamethasone sodium phosphate in female mice was 794 mg/kg.
-
DOSAGE AND ADMINISTRATION
Dexamethasone sodium phosphate injection, 10 mg/mL– For intravenous and intramuscular injection only.
Dexamethasone sodium phosphate injection can be given directly from the vial, or it can be added to Sodium Chloride Injection or Dextrose Injection and administered by intravenous drip.
Solutions used for intravenous administration or further dilution of this product should be preservative free when used in the neonate, especially the premature infant.
When it is mixed with an infusion solution, sterile precautions should be observed. Since infusion solutions generally do not contain preservatives, mixtures should be used within 24 hours.
DOSAGE REQUIREMENTS ARE VARIABLE AND MUST BE INDIVIDUALIZED ON THE BASIS OF THE DISEASE AND THE RESPONSE OF THE PATIENT.
Intravenous and Intramuscular Injection
The initial dosage of dexamethasone sodium phosphate injection varies from 0.5 to 9 mg a day depending on the disease being treated. In less severe diseases doses lower than 0.5 mg may suffice, while in severe diseases doses higher than 9 mg may be required.
The initial dosage should be maintained or adjusted until the patient’s response is satisfactory. If a satisfactory clinical response does not occur after a reasonable period of time, discontinue dexamethasone sodium phosphate injection and transfer the patient to other therapy.
After a favorable initial response, the proper maintenance dosage should be determined by decreasing the initial dosage in small amounts to the lowest dosage that maintains an adequate clinical response.
Patients should be observed closely for signs that might require dosage adjustment, including changes in clinical status resulting from remissions or exacerbations of the disease, individual drug responsiveness, and the effect of stress (e.g., surgery, infection, trauma). During stress it may be necessary to increase dosage temporarily.
If the drug is to be stopped after more than a few days of treatment, it usually should be withdrawn gradually.
When the intravenous route of administration is used, dosage usually should be the same as the oral dosage. In certain overwhelming, acute, life-threatening situations, however, administration in dosages exceeding the usual dosages may be justified and may be in multiples of the oral dosages. The slower rate of absorption by intramuscular administration should be recognized.
Shock
There is a tendency in current medical practice to use high (pharmacologic) doses of corticosteroids for the treatment of unresponsive shock. The following dosages of dexamethasone sodium phosphate injection have been suggested by various authors:
Author
Dosage
Cavanagh 1
3 mg/kg of body weight per 24 hours by constant intravenous infusion after an initial intravenous injection of 20 mg
Dietzman 2
2 to 6 mg/kg of body weight as a single intravenous injection
Frank 3
40 mg initially followed by repeat intravenous injection every 4 to 6 hours while shock persists
Oaks 4
40 mg initially followed by repeat intravenous injection every 2 to 6 hours while shock persists
Schumer 5
1 mg/kg of body weight as a single intravenous injection
Administration of high dose corticosteroid therapy should be continued only until the patient’s condition has stabilized and usually not longer than 48 to 72 hours.
Although adverse reactions associated with high dose, short-term corticosteroid therapy are uncommon, peptic ulceration may occur.
Cerebral Edema
Dexamethasone sodium phosphate injection is generally administered initially in a dosage of 10 mg intravenously followed by four mg every six hours intramuscularly until the symptoms of cerebral edema subside. Response is usually noted within 12 to 24 hours and dosage may be reduced after two to four days and gradually discontinued over a period of five to seven days. For palliative management of patients with recurrent or inoperable brain tumors, maintenance therapy with 2 mg two or three times a day may be effective.
Acute Allergic Disorders
In acute, self-limited allergic disorders or acute exacerbations of chronic allergic disorders, the following dosage schedule combining parenteral and oral therapy is suggested:
Dexamethasone sodium phosphate injection, first day, 4 or 8 mg intramuscularly.
Dexamethasone tablets, 0.75 mg: second and third days, 4 tablets in two divided doses each day; fourth day, 2 tablets in two divided doses; fifth and sixth days, 1 tablet each day; seventh day, no treatment; eighth day, follow-up visit.
This schedule is designed to ensure adequate therapy during acute episodes, while minimizing the risk of overdosage in chronic cases.
Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration, whenever the solution and container permit.
-
HOW SUPPLIED
Dexamethasone Sodium Phosphate Injection, USP (Preservative Free) equivalent to 10 mg dexamethasone phosphate, is supplied in a single dose vial as follows:
Product
No.
NDC
No.
Strength
Vial Size
500601
63323-506-01
10 mg per mL
1 mL vial, packaged in twenty-fives.
This container closure is not made with natural rubber latex.
-
REFERENCES
- Cavanagh, D.; Singh, K.B.: Endotoxin shock in pregnancy and abortion, in: “Corticosteroids in the Treatment of Shock”, Schumer, W.; Nyhus, L.M., Editors, Urbana, University of Illinois Press, 1970, pp. 86-96.
- Dietzman, R.H.; Ersek, R.A.; Bloch, J.M.; Lilleheir, R.C.: High-output, low-resistance gram-negative septic shock in man, Angiology 20: 691-700, Dec. 1969.
- Frank, E.: Clinical observations in shock and management (in: Shields, T.F., ed.: Symposium on current concepts and management of shock), J. Maine Med. Ass. 59: 195-200, Oct. 1968.
- Oaks, W. W.; Cohen, H.E.: Endotoxin shock in the geriatric patient, Geriat. 22: 120-130, Mar. 1967.
- Schumer, W.; Nyhus, L.M.: Corticosteroid effect on biochemical parameters of human oligemic shock, Arch. Surg. 100: 405-408, Apr. 1970.
- SPL UNCLASSIFIED SECTION
- SPL UNCLASSIFIED SECTION
-
DESCRIPTION
Lidocaine hydrochloride injection, USP is sterile, nonpyrogenic, aqueous solution that contains a local anesthetic agent and is administered parenterally by injection. See INDICATIONS AND USAGE section for specific uses.
Lidocaine hydrochloride injection, USP contains lidocaine hydrochloride, which is chemically designated as acetamide, 2-(diethylamino)-N-(2,6-dimethylphenyl)-, monohydrochloride and has the molecular weight 270.8. Lidocaine hydrochloride (C 14H 22N 2O • HCl) has the following structural formula: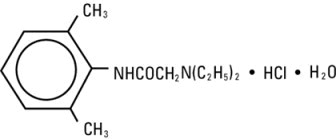
Lidocaine hydrochloride injection, USP is a sterile, nonpyrogenic, isotonic solution containing sodium chloride. The pH of the solution is adjusted to approximately 6.5 (5.0 to 7.0) with sodium hydroxide and/or hydrochloric acid.
-
CLINICAL PHARMACOLOGY
Mechanism of Action
Lidocaine stabilizes the neuronal membrane by inhibiting the ionic fluxes required for the initiation and conduction of impulses thereby effecting local anesthetic action.
Hemodynamics
Excessive blood levels may cause changes in cardiac output, total peripheral resistance, and mean arterial pressure. With central neural blockade these changes may be attributable to block of autonomic fibers, a direct depressant effect of the local anesthetic agent on various components of the cardiovascular system, and/or the beta-adrenergic receptor stimulating action of epinephrine when present. The net effect is normally a modest hypotension when the recommended dosages are not exceeded.
Pharmacokinetics and Metabolism
Information derived from diverse formulations, concentrations and usages reveals that lidocaine is completely absorbed following parenteral administration, its rate of absorption depending, for example, upon various factors such as the site of administration and the presence or absence of a vasoconstrictor agent. Except for intravascular administration, the highest blood levels are obtained following intercostal nerve block and the lowest after subcutaneous administration.
The plasma binding of lidocaine is dependent on drug concentration, and the fraction bound decreases with increasing concentration. At concentrations of 1 to 4 mcg of free base per mL 60 to 80 percent of lidocaine is protein bound. Binding is also dependent on the plasma concentration of the alpha-1-acid glycoprotein.
Lidocaine crosses the blood-brain and placental barriers, presumably by passive diffusion.
Lidocaine is metabolized rapidly by the liver, and metabolites and unchanged drug are excreted by the kidneys. Biotransformation includes oxidative N-dealkylation, ring hydroxylation, cleavage of the amide linkage, and conjugation. N-dealkylation, a major pathway of biotransformation, yields the metabolites monoethylglycinexylidide and glycinexylidide. The pharmacological/toxicological actions of these metabolites are similar to, but less potent than, those of lidocaine. Approximately 90% of lidocaine administered is excreted in the form of various metabolites, and less than 10% is excreted unchanged. The primary metabolite in urine is a conjugate of 4-hydroxy-2,6-dimethylaniline.
The elimination half-life of lidocaine following an intravenous bolus injection is typically 1.5 to 2 hours. Because of the rapid rate at which lidocaine is metabolized, any condition that affects liver function may alter lidocaine kinetics. The half-life may be prolonged two-fold or more in patients with liver dysfunction. Renal dysfunction does not affect lidocaine kinetics but may increase the accumulation of metabolites.
Factors such as acidosis and the use of CNS stimulants and depressants affect the CNS levels of lidocaine required to produce overt systemic effects. Objective adverse manifestations become increasingly apparent with increasing venous plasma levels above 6 mcg free base per mL. In the rhesus monkey arterial blood levels of 18 to 21 mcg/mL have been shown to be threshold for convulsive activity. -
INDICATIONS AND USAGE
Lidocaine hydrochloride injection is indicated for production of local or regional anesthesia by infiltration techniques such as percutaneous injection and intravenous regional anesthesia by peripheral nerve block techniques such as brachial plexus and intercostal and by central neural techniques such as lumbar and caudal epidural blocks, when the accepted procedures for these techniques as described in standard textbooks are observed.
- CONTRAINDICATIONS
-
WARNINGS
LIDOCAINE HYDROCHLORIDE INJECTION FOR INFILTRATION AND NERVE BLOCK SHOULD BE EMPLOYED ONLY BY CLINICIANS WHO ARE WELL VERSED IN DIAGNOSIS AND MANAGEMENT OF DOSE-RELATED TOXICITY AND OTHER ACUTE EMERGENCIES THAT MIGHT ARISE FROM THE BLOCK TO BE EMPLOYED AND THEN ONLY AFTER ENSURING THE IMMEDIATE AVAILABILITY OF OXYGEN, OTHER RESUSCITATIVE DRUGS, CARDIOPULMONARY EQUIPMENT AND THE PERSONNEL NEEDED FOR PROPER MANAGEMENT OF TOXIC REACTIONS AND RELATED EMERGENCIES (see also ADVERSE REACTIONSand PRECAUTIONS). DELAY IN PROPER MANAGEMENT OF DOSE-RELATED TOXICITY, UNDERVENTILATION FROM ANY CAUSE AND/OR ALTERED SENSITIVITY MAY LEAD TO THE DEVELOPMENT OF ACIDOSIS, CARDIAC ARREST AND, POSSIBLY, DEATH.
Methemoglobinemia
Cases of methemoglobinemia have been reported in association with local anesthetic use. Although all patients are at risk for methemoglobinemia, patients with glucose-6-phosphate dehydrogenase deficiency, congenital or idiopathic methemoglobinemia, cardiac or pulmonary compromise, infants under 6 months of age, and concurrent exposure to oxidizing agents or their metabolites are more susceptible to developing clinical manifestations of the condition. If local anesthetics must be used in these patients, close monitoring for symptoms and signs of methemoglobinemia is recommended.
Signs of methemoglobinemia may occur immediately or may be delayed some hours after exposure, and are characterized by a cyanotic skin discoloration and/or abnormal coloration of the blood. Methemoglobin levels may continue to rise; therefore, immediate treatment is required to avert more serious central nervous system and cardiovascular adverse effects, including seizures, coma, arrhythmias, and death. Discontinue lidocaine hydrochloride and any other oxidizing agents. Depending on the severity of the signs and symptoms, patients may respond to supportive care, i.e., oxygen therapy, hydration. A more severe clinical presentation may require treatment with methylene blue, exchange transfusion, or hyperbaric oxygen.
Intra-articular infusions of local anesthetics following arthroscopic and other surgical procedures is an unapproved use, and there have been postmarketing reports of chondrolysis in patients receiving such infusions. The majority of reported cases of chondrolysis have involved the shoulder joint; cases of gleno-humeral chondrolysis have been described in pediatric and adult patients following intra-articular infusions of local anesthetics with and without epinephrine for periods of 48 to 72 hours. There is insufficient information to determine whether shorter infusion periods are not associated with these findings. The time of onset of symptoms, such as joint pain, stiffness and loss of motion can be variable, but may begin as early as the 2 nd month after surgery. Currently, there is no effective treatment for chondrolysis; patients who experienced chondrolysis have required additional diagnostic and therapeutic procedures and some required arthroplasty or shoulder replacement.
To avoid intravascular injection, aspiration should be performed before the local anesthetic solution is injected. The needle must be repositioned until no return of blood can be elicited by aspiration. Note, however, that the absence of blood in the syringe does not guarantee that intravascular injection has been avoided.
Anaphylactic reactions may occur following administration of lidocaine hydrochloride (see ADVERSE REACTIONS).
In the case of severe reaction, discontinue the use of the drug. -
PRECAUTIONS
General
The safety and effectiveness of lidocaine hydrochloride depend on proper dosage, correct technique, adequate precautions, and readiness for emergencies. Standard textbooks should be consulted for specific techniques and precautions for various regional anesthetic procedures.
Resuscitative equipment, oxygen, and other resuscitative drugs should be available for immediate use (see WARNINGSand ADVERSE REACTIONS). The lowest dosage that results in effective anesthesia should be used to avoid high plasma levels and serious adverse effects. Syringe aspirations should also be performed before and during each supplemental injection when using indwelling catheter techniques. During the administration of epidural anesthesia, it is recommended that a test dose be administered initially and that the patient be monitored for central nervous system toxicity and cardiovascular toxicity, as well as for signs of unintended intrathecal administration, before proceeding. When clinical conditions permit, consideration should be given to employing local anesthetic solutions that contain epinephrine for the test dose because circulatory changes compatible with epinephrine may also serve as a warning sign of unintended intravascular injection. An intravascular injection is still possible even if aspirations for blood are negative. Repeated doses of lidocaine hydrochloride may cause significant increases in blood levels with each repeated dose because of slow accumulation of the drug or its metabolites. Tolerance to elevated blood levels varies with the status of the patient. Debilitated, elderly patients, acutely ill patients, and children should be given reduced doses commensurate with their age and physical condition. Lidocaine hydrochloride should also be used with caution in patients with severe shock or heart block.
Lumbar and caudal epidural anesthesia should be used with extreme caution in persons with the following conditions: existing neurological disease, spinal deformities, septicemia, and severe hypertension.
Local anesthetic solutions containing a vasoconstrictor should be used cautiously and in carefully circumscribed quantities in areas of the body supplied by end arteries or having otherwise compromised blood supply. Patients with peripheral vascular disease and those with hypertensive vascular disease may exhibit exaggerated vasoconstrictor response. Ischemic injury or necrosis may result. Preparations containing a vasoconstrictor should be used with caution in patients during or following the administration of potent general anesthetic agents, since cardiac arrhythmias may occur under such conditions.
Careful and constant monitoring of cardiovascular and respiratory (adequacy of ventilation) vital signs and the patient’s state of consciousness should be accomplished after each local anesthetic injection. It should be kept in mind at such times that restlessness, anxiety, tinnitus, dizziness, blurred vision, tremors, depression or drowsiness may be early warning signs of central nervous system toxicity.
Since amide-type local anesthetics are metabolized by the liver, lidocaine hydrochloride injection should be used with caution in patients with hepatic disease. Patients with severe hepatic disease, because of their inability to metabolize local anesthetics normally, are at greater risk of developing toxic plasma concentrations. Lidocaine hydrochloride injection should also be used with caution in patients with impaired cardiovascular function since they may be less able to compensate for functional changes associated with the prolongation of A-V conduction produced by these drugs.
Many drugs used during the conduct of anesthesia are considered potential triggering agents for familial malignant hyperthermia. Since it is not known whether amide-type local anesthetics may trigger this reaction and since the need for supplemental general anesthesia cannot be predicted in advance, it is suggested that a standard protocol for the management of malignant hyperthermia should be available. Early unexplained signs of tachycardia, tachypnea, labile blood pressure and metabolic acidosis may precede temperature elevation. Successful outcome is dependent on early diagnosis, prompt discontinuance of the suspect triggering agent(s) and institution of treatment, including oxygen therapy, indicated supportive measures and dantrolene (consult dantrolene sodium intravenous package insert before using).
Proper tourniquet technique, as described in publications and standard textbooks, is essential in the performance of intravenous regional anesthesia. Solutions containing epinephrine or other vasoconstrictors should not be used for this technique.
Lidocaine hydrochloride should be used with caution in persons with known drug sensitivities. Patients allergic to para-aminobenzoic acid derivatives (procaine, tetracaine, benzocaine, etc.) have not shown cross sensitivity to lidocaine hydrochloride.Use in the Head and Neck Area
Small doses of local anesthetics injected into the head and neck area, including retrobulbar, dental and stellate ganglion blocks, may produce adverse reactions similar to systemic toxicity seen with unintentional intravascular injections of larger doses. Confusion, convulsions, respiratory depression and/or respiratory arrest, and cardiovascular stimulation or depression have been reported. These reactions may be due to intra-arterial injection of the local anesthetic with retrograde flow to the cerebral circulation. Patients receiving these blocks should have their circulation and respiration monitored and be constantly observed. Resuscitative equipment and personnel for treating adverse reactions should be immediately available. Dosage recommendations should not be exceeded (see DOSAGE AND ADMINISTRATION).
Information for Patients
When appropriate, patients should be informed in advance that they may experience temporary loss of sensation and motor activity, usually in the lower half of the body, following proper administration of epidural anesthesia.
Inform patients that use of local anesthetics may cause methemoglobinemia, a serious condition that must be treated promptly. Advise patients or caregivers to seek immediate medical attention if they or someone in their care experience the following signs or symptoms: pale, gray, or blue colored skin (cyanosis); headache; rapid heart rate; shortness of breath; lightheadedness; or fatigue.Clinically Significant Drug Interactions
The administration of local anesthetic solutions containing epinephrine or norepinephrine to patients receiving monoamine oxidase inhibitors or tricyclic antidepressants may produce severe, prolonged hypertension.
Phenothiazines and butyrophenones may reduce or reverse the pressor effect of epinephrine.
Concurrent use of these agents should generally be avoided. In situations when concurrent therapy is necessary, careful patient monitoring is essential.
Concurrent administration of vasopressor drugs (for the treatment of hypotension related to obstetric blocks) and ergot-type oxytocic drugs may cause severe, persistent hypertension or cerebrovascular accidents.Drug/Laboratory Test Interactions
The intramuscular injection of lidocaine hydrochloride may result in an increase in creatine phosphokinase levels. Thus, the use of this enzyme determination, without isoenzyme separation, as a diagnostic test for the presence of acute myocardial infarction may be compromised by the intramuscular injection of lidocaine hydrochloride.
Patients who are administered local anesthetics are at increased risk of developing methemoglobinemia when concurrently exposed to the following drugs, which could include other local anesthetics:
Examples of Drugs Associated with Methemoglobinemia:
Class
Examples
Nitrates/Nitrites
nitric oxide, nitroglycerin, nitroprusside, nitrous oxide
Local anesthetics
articaine, benzocaine, bupivacaine, lidocaine, mepivacaine, prilocaine, procaine, ropivacaine, tetracaine
Antineoplastic agents
cyclophosphamide, flutamide, hydroxyurea, ifosfamide, rasburicase
Antibiotics
dapsone, nitrofurantoin, para-aminosalicylic acid, sulfonamides
Antimalarials
chloroquine, primaquine
Anticonvulsants
Phenobarbital, phenytoin, sodium valproate
Other drugs
acetaminophen, metoclopramide, quinine,
sulfasalazine
Carcinogenesis, Mutagenesis, Impairment of Fertility
Studies of lidocaine hydrochloride in animals to evaluate the carcinogenic and mutagenic potential or the effect on fertility have not been conducted.
Pregnancy
Teratogenic Effects
Reproduction studies have been performed in rats at doses up to 6.6 times the human dose and have revealed no evidence of harm to the fetus caused by lidocaine hydrochloride. There are, however, no adequate and well-controlled studies in pregnant women. Animal reproduction studies are not always predictive of human response. General consideration should be given to this fact before administering lidocaine hydrochloride to women of childbearing potential, especially during early pregnancy when maximum organogenesis takes place.
Labor and Delivery
Local anesthetics rapidly cross the placenta and when used for epidural, paracervical, pudendal or caudal block anesthesia, can cause varying degrees of maternal, fetal and neonatal toxicity see CLINICAL PHARMACOLOGY, Pharmacokinetics and Metabolism). The potential for toxicity depends upon the procedure performed, the type and amount of drug used, and the technique of drug administration. Adverse reactions in the parturient, fetus and neonate involve alterations of the central nervous system, peripheral vascular tone and cardiac function.
Maternal hypotension has resulted from regional anesthesia. Local anesthetics produce vasodilation by blocking sympathetic nerves. Elevating the patient’s legs and positioning her on her left side will help prevent decreases in blood pressure.
The fetal heart rate also should be monitored continuously, and electronic fetal monitoring is highly advisable.
Epidural, spinal, paracervical, or pudendal anesthesia may alter the forces of parturition through changes in uterine contractility or maternal expulsive efforts. In one study, paracervical block anesthesia was associated with a decrease in the mean duration of first stage labor and facilitation of cervical dilation. However, spinal and epidural anesthesia have also been reported to prolong the second stage of labor by removing the parturient’s reflex urge to bear down or by interfering with motor function. The use of obstetrical anesthesia may increase the need for forceps assistance.
The use of some local anesthetic drug products during labor and delivery may be followed by diminished muscle strength and tone for the first day or two of life. The long-term significance of these observations is unknown. Fetal bradycardia may occur in 20 to 30 percent of patients receiving paracervical nerve block anesthesia with the amide-type local anesthetics and may be associated with fetal acidosis. Fetal heart rate should always be monitored during paracervical anesthesia. The physician should weigh the possible advantages against risks when considering a paracervical block in prematurity, toxemia of pregnancy, and fetal distress. Careful adherence to recommended dosage is of the utmost importance in obstetrical paracervical block. Failure to achieve adequate analgesia with recommended doses should arouse suspicion of intravascular or fetal intracranial injection. Cases compatible with unintended fetal intracranial injection of local anesthetic solution have been reported following intended paracervical or pudendal block or both. Babies so affected present with unexplained neonatal depression at birth, which correlates with high local anesthetic serum levels, and often manifest seizures within six hours. Prompt use of supportive measures combined with forced urinary excretion of the local anesthetic has been used successfully to manage this complication.
Case reports of maternal convulsions and cardiovascular collapse following use of some local anesthetics for paracervical block in early pregnancy (as anesthesia for elective abortion) suggest that systemic absorption under these circumstances may be rapid. The recommended maximum dose of each drug should not be exceeded. Injection should be made slowly and with frequent aspiration. Allow a 5-minute interval between sides.Nursing Mothers
It is not known whether this drug is excreted in human milk. Because many drugs are excreted in human milk, caution should be exercised when lidocaine hydrochloride is administered to a nursing woman.
Pediatric Use
Dosages in children should be reduced, commensurate with age, body weight and physical condition, see DOSAGE AND ADMINISTRATION.
-
ADVERSE REACTIONS
Systemic
Adverse experiences following the administration of lidocaine hydrochloride are similar in nature to those observed with other amide local anesthetic agents. These adverse experiences are, in general, dose-related and may result from high plasma levels caused by excessive dosage, rapid absorption or inadvertent intravascular injection, or may result from a hypersensitivity, idiosyncrasy or diminished tolerance on the part of the patient. Serious adverse experiences are generally systemic in nature. The following types are those most commonly reported:
Central Nervous System
CNS manifestations are excitatory and/or depressant and may be characterized by lightheadedness, nervousness, apprehension, euphoria, confusion, dizziness, drowsiness, tinnitus, blurred or double vision, vomiting, sensations of heat, cold or numbness, twitching, tremors, convulsions, unconsciousness, respiratory depression and arrest. The excitatory manifestations may be very brief or may not occur at all, in which case the first manifestation of toxicity may be drowsiness merging into unconsciousness and respiratory arrest.
Drowsiness following the administration of lidocaine hydrochloride is usually an early sign of a high blood level of the drug and may occur as a consequence of rapid absorption.
Cardiovascular System
Cardiovascular manifestations are usually depressant and are characterized by bradycardia, hypotension, and cardiovascular collapse, which may lead to cardiac arrest.
Allergic
Allergic reactions are characterized by cutaneous lesions, urticaria, edema or anaphylactoid reactions. Allergic reactions may occur as a result of sensitivity to local anesthetic agents. Allergic reactions, including anaphylactic reactions, may occur as a result of sensitivity to lidocaine, but are infrequent. If allergic reactions do occur, they should be managed by conventional means. The detection of sensitivity by skin testing is of doubtful value.
There have been no reports of cross sensitivity between lidocaine hydrochloride and procainamide or between lidocaine hydrochloride and quinidine.Neurologic
The incidences of adverse reactions associated with the use of local anesthetics may be related to the total dose of local anesthetic administered and are also dependent upon the particular drug used, the route of administration and the physical status of the patient. In a prospective review of 10,440 patients who received lidocaine hydrochloride for spinal anesthesia, the incidences of adverse reactions were reported to be about 3 percent each for positional headaches, hypotension and backache; 2 percent for shivering; and less than 1 percent each for peripheral nerve symptoms, nausea, respiratory inadequacy and double vision. Many of these observations may be related to local anesthetic techniques, with or without a contribution from the local anesthetic.
In the practice of caudal or lumbar epidural block, occasional unintentional penetration of the subarachnoid space by the catheter may occur. Subsequent adverse effects may depend partially on the amount of drug administered subdurally. These may include spinal block of varying magnitude (including total spinal block), hypotension secondary to spinal block, loss of bladder and bowel control, and loss of perineal sensation and sexual function. Persistent motor, sensory and/or autonomic (sphincter control) deficit of some lower spinal segments with slow recovery (several months) or incomplete recovery have been reported in rare instances when caudal or lumbar epidural block has been attempted. Backache and headache have also been noted following use of these anesthetic procedures.
There have been reported cases of permanent injury to extraocular muscles requiring surgical repair following retrobulbar administration. -
OVERDOSAGE
Acute emergencies from local anesthetics are generally related to high plasma levels encountered during therapeutic use of local anesthetics or to unintended subarachnoid injection of local anesthetic solution (see ADVERSE REACTIONS, WARNINGS, and PRECAUTIONS).
Management of Local Anesthetic Emergencies
The first consideration is prevention, best accomplished by careful and constant monitoring of cardiovascular and respiratory vital signs and the patient’s state of consciousness after each local anesthetic injection. At the first sign of change, oxygen should be administered.
The first step in the management of convulsions, as well as underventilation or apnea due to unintended subarachnoid injection of drug solution, consists of immediate attention to the maintenance of a patent airway and assisted or controlled ventilation with oxygen and a delivery system capable of permitting immediate positive airway pressure by mask. Immediately after the institution of these ventilatory measures, the adequacy of the circulation should be evaluated, keeping in mind that drugs used to treat convulsions sometimes depress the circulation when administered intravenously. Should convulsions persist despite adequate respiratory support, and if the status of the circulation permits, small increments of an ultra-short acting barbiturate (such as thiopental or thiamylal) or a benzodiazepine (such as diazepam) may be administered intravenously. The clinician should be familiar, prior to the use of local anesthetics, with these anticonvulsant drugs. Supportive treatment of circulatory depression may require administration of intravenous fluids and, when appropriate, a vasopressor as directed by the clinical situation (e.g., ephedrine).
If not treated immediately, both convulsions and cardiovascular depression can result in hypoxia, acidosis, bradycardia, arrhythmias and cardiac arrest. Underventilation or apnea due to unintentional subarachnoid injection of local anesthetic solution may produce these same signs and also lead to cardiac arrest if ventilatory support is not instituted. If cardiac arrest should occur, standard cardiopulmonary resuscitative measures should be instituted.
Endotracheal intubation, employing drugs and techniques familiar to the clinician, may be indicated, after initial administration of oxygen by mask, if difficulty is encountered in the maintenance of a patent airway or if prolonged ventilatory support (assisted or controlled) is indicated.
Dialysis is of negligible value in the treatment of acute overdosage with lidocaine hydrochloride.
The oral LD 50 of lidocaine hydrochloride in non-fasted female rats is 459 (346 to 773) mg/kg (as the salt) and 214 (159 to 324) mg/kg (as the salt) in fasted female rats. -
DOSAGE AND ADMINISTRATION
Table 1 (Recommended Dosages) summarizes the recommended volumes and concentrations of lidocaine hydrochloride injection for various types of anesthetic procedures. The dosages suggested in this table are for normal healthy adults and refer to the use of epinephrine-free solutions. When larger volumes are required, only solutions containing epinephrine should be used except in those cases where vasopressor drugs may be contraindicated.
There have been adverse event reports of chondrolysis in patients receiving intra-articular infusions of local anesthetics following arthroscopic and other surgical procedures. Lidocaine hydrochloride injection is not approved for this use (see WARNINGSand DOSAGE AND ADMINISTRATION).
These recommended doses serve only as a guide to the amount of anesthetic required for most routine procedures. The actual volumes and concentrations to be used depend on a number of factors such as type and extent of surgical procedure, depth of anesthesia and degree of muscular relaxation required, duration of anesthesia required, and the physical condition of the patient. In all cases the lowest concentration and smallest dose that will produce the desired result should be given. Dosages should be reduced for children and for the elderly and debilitated patients and patients with cardiac and/or liver disease.
The onset of anesthesia, the duration of anesthesia and the degree of muscular relaxation are proportional to the volume and concentration (i.e., total dose) of local anesthetic used. Thus, an increase in volume and concentration of lidocaine hydrochloride injection will decrease the onset of anesthesia, prolong the duration of anesthesia, provide a greater degree of muscular relaxation and increase the segmental spread of anesthesia. However, increasing the volume and concentration of lidocaine hydrochloride injection may result in a more profound fall in blood pressure when used in epidural anesthesia. Although the incidence of side effects with lidocaine hydrochloride is quite low, caution should be exercised when employing large volumes and concentrations, since the incidence of side effects is directly proportional to the total dose of local anesthetic agent injected.Epidural Anesthesia
For epidural anesthesia, only the following dosage forms of lidocaine hydrochloride injection are recommended:
1% without epinephrine 5 mL ampules
2% without epinephrine 2 mL ampules
Although these solutions are intended specifically for epidural anesthesia, they may also be used for infiltration and peripheral nerve block, provided they are employed as single dose units. These solutions contain no bacteriostatic agent.
In epidural anesthesia, the dosage varies with the number of dermatomes to be anesthetized (generally 2 to 3 mL of the indicated concentration per dermatome).Caudal and Lumbar Epidural Block
As a precaution against the adverse experience sometimes observed following unintentional penetration of the subarachnoid space, a test dose such as 2 to 3 mL of 1.5% lidocaine hydrochloride should be administered at least 5 minutes prior to injecting the total volume required for a lumbar or caudal epidural block. The test dose should be repeated if the patient is moved in a manner that may have displaced the catheter. Epinephrine, if contained in the test dose (10 to 15 mcg have been suggested), may serve as a warning of unintentional intravascular injection. If injected into a blood vessel, this amount of epinephrine is likely to produce a transient “epinephrine response” within 45 seconds, consisting of an increase in heart rate and systolic blood pressure, circumoral pallor, palpitations and nervousness in the unsedated patient. The sedated patient may exhibit only a pulse rate increase of 20 or more beats per minute for 15 or more seconds. Patients on beta blockers may not manifest changes in heart rate, but blood pressure monitoring can detect an evanescent rise in systolic blood pressure. Adequate time should be allowed for onset of anesthesia after administration of each test dose. The rapid injection of a large volume of lidocaine hydrochloride injection through the catheter should be avoided, and, when feasible, fractional doses should be administered.
In the event of the known injection of a large volume of local anesthetic solution into the subarachnoid space, after suitable resuscitation and if the catheter is in place, consider attempting the recovery of drug by draining a moderate amount of cerebrospinal fluid (such as 10 mL) through the epidural catheter. -
MAXIMUM RECOMMENDED DOSAGES
Adults
For normal healthy adults, the maximum individual dose should not exceed 4.5 mg/kg (2 mg/lb) of body weight, and in general it is recom mended that the max i m um t otal dose does not exceed 300 mg. For continuous epidural or caudal anesthesia, the maximum recommended dosage should not be administered at intervals of less than 90 minutes. When continuous lumbar or caudal epidural anesthesia is used for non-obstetrical procedures, more drug may be administered if required to produce adequate anesthesia.
The maximum recommended dose per 90 minute period of lidocaine hydrochloride for paracervical block in obstetrical patients and non-obstetrical patients is 200 mg total. One half of the total dose is usually administered to each side. Inject slowly, five minutes between sides (see also discussion of paracervical block in PRECAUTIONS).
For intravenous regional anesthesia, the dose administered should not exceed 4 mg/kg in adults.Children
It is difficult to recommend a maximum dose of any drug for children, since this varies as a function of age and weight. For children over 3 years of age who have a normal lean body mass and normal body development, the maximum dose is determined by the child’s age and weight. For example, in a child of 5 years weighing 50 lbs the dose of lidocaine hydrochloride should not exceed 75 to 100 mg (1.5 to 2 mg/lb). The use of even more dilute solutions (i.e., 0.25 to 0.5%) and total dosages not to exceed 3 mg/kg (1.4 mg/lb) are recommended for induction of intravenous regional anesthesia in children.
In order to guard against systemic toxicity, the lowest effective concentration and lowest effective dose should be used at all times. In some cases it will be necessary to dilute available concentrations with 0.9% sodium chloride injection in order to obtain the required final concentration.
NOTE: Parenteral drug products should be inspected visually for particulate matter and discoloration prior to administration whenever the solution and container permit. Do not use if solution is discolored or contains a precipitate.Table 1: Recommended Dosages Procedure Lidocaine Hydrochloride Injection
(without epinephrine)Conc (%) Vol (mL) Total Dose (mg) *Dose determined by number of dermatomes to be anesthetized (2 to 3 mL/dermatome).
Infiltration
Percutaneous
0.5 or 1
1 to 60
5 to 300
Intravenous regional
0.5
10 to 60
50 to 300
Peripheral Nerve Blocks, e.g.,
Brachial
1.5
15 to 20
225 to 300
Dental
2
1 to 5
20 to 100
Intercostal
1
3
30
Paravertebral
1
3 to 5
30 to 50
Pudendal (each side)
1
10
100
Paracervical
Obstetrical analgesia (each side)
1
10
100
Sympathetic Nerve Blocks, e.g.,
Cervical (stellate ganglion)
1
5
50
Lumbar
1
5 to 10
50 to 100
Central Neural Blocks
Epidural*
Thoracic
1
20 to 30
200 to 300
Lumbar
Analgesia
1
25 to 30
250 to 300
Anesthesia
1.5
15 to 20
225 to 300
2
10 to 15
200 to 300
Caudal
Obstetrical analgesia
1
20 to 30
200 to 300
Surgical anesthesia
1.5
15 to 20
225 to 300
THE ABOVE SUGGESTED CONCENTRATIONS AND VOLUMES SERVE ONLY AS A GUIDE. OTHER VOLUMES AND CONCENTRATIONS MAY BE USED PROVIDED THE TOTAL MAXIMUM RECOMMENDED DOSE IS NOT EXCEEDED.
- STERILIZATION, STORAGE AND TECHNICAL PROCEDURES
-
HOW SUPPLIED
Lidocaine Hydrochloride Injection USP, is supplied as follows:
Lidocaine Hydrochloride Injection USP, 1% (10 mg/mL)
2 mL Ampules in a Carton of 10 NDC 55150-158-72
Sterile, Nonpyrogenic
Discard unused portion.
Store at 20° to 25°C (68° to 77°F) [see USP Controlled Room Temperature].
Distributed by:
AuroMedics Pharma LLC
279 Princeton-Hightstown Rd.
E. Windsor, NJ 08520
Manufactured by:
Eugia Pharma Specialities Limited
Hyderabad - 500032
India
Revised: October 2021 - SPL UNCLASSIFIED SECTION
- ACTIVE INGREDIENT
- Purpose:
- Warnings:
- Do not use:
- Ask a doctor before use if you have:
- Stop Use:
- Keep Out Of Reach Of Children
- Directions Povidone iodine:
- Other information:
- INDICATIONS & USAGE
- Inactive Ingredients
- SPL UNCLASSIFIED SECTION
- Active ingredient
- Purpose
- Uses
-
Warnings
For external use only
Flammable - keep away from fire or flame
When using this product do not
- get into eyes
- apply over large areas of the body
- in case of deep or puncture wounds, animal bites or serious burns consult a doctor
- Directions
- Other information
- Inactive ingredient
-
PRINCIPAL DISPLAY PANEL
PRNCIPAL DISPLAY PANEL
NDC: 76420-767-01
RX Only
Dexlido™
Kit Contains
1 Dexamethasone Sodium Phosphate Inj., USP 10mg/mL (1mL)
1 Lidocaine HCl Injection, USP 1% Ampule (2mL)
1 Povidone-Iodine Swabsticks (3 Swabs)
2 Isopropyl Alcohol 70% Prep Pads
1 Pair Nitrile Powder Free Sterile Gloves (M)
1 Drape
1 Adhesive Bandage
5 Non Sterile 4x4 Gauze
Needles and Syringes Not Included
1 Dose
Single Use Only
Distributed by:
Enovachem™
PHARMACEUTICALS
Torrance, CA 90501

-
INGREDIENTS AND APPEARANCE
DEXLIDO KIT
dexamethasone sodium phosphate, lidocaine hydrochloride, povidine iodine kitProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:76420-767 Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:76420-767-01 1 in 1 CARTON; Type 1: Convenience Kit of Co-Package 05/23/2016 Quantity of Parts Part # Package Quantity Total Product Quantity Part 1 1 VIAL 1 mL Part 2 1 AMPULE 2 mL Part 3 1 PACKET 0.9 mL Part 4 2 POUCH 10 mL Part 1 of 4 DEXAMETHASONE SODIUM PHOSPHATE
dexamethasone sodium phosphate injection, solutionProduct Information Item Code (Source) NDC:63323-506 Route of Administration INTRAMUSCULAR, INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DEXAMETHASONE SODIUM PHOSPHATE (UNII: AI9376Y64P) (DEXAMETHASONE - UNII:7S5I7G3JQL) DEXAMETHASONE PHOSPHATE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CITRATE (UNII: 1Q73Q2JULR) CITRIC ACID MONOHYDRATE (UNII: 2968PHW8QP) SODIUM HYDROXIDE (UNII: 55X04QC32I) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:63323-506-01 25 in 1 TRAY 1 1 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA040491 05/29/2003 Part 2 of 4 LIDOCAINE HYDROCHLORIDE
lidocaine hydrochloride injection, solutionProduct Information Item Code (Source) NDC:55150-158 Route of Administration INFILTRATION, PERINEURAL, INTRACAUDAL, EPIDURAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength LIDOCAINE HYDROCHLORIDE (UNII: V13007Z41A) (LIDOCAINE - UNII:98PI200987) LIDOCAINE HYDROCHLORIDE ANHYDROUS 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength SODIUM CHLORIDE (UNII: 451W47IQ8X) 7 mg in 1 mL WATER (UNII: 059QF0KO0R) SODIUM HYDROXIDE (UNII: 55X04QC32I) HYDROCHLORIC ACID (UNII: QTT17582CB) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:55150-158-72 10 in 1 CARTON 1 2 mL in 1 AMPULE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date ANDA ANDA203040 03/14/2013 Part 3 of 4 POVIDINE IODINE
povidine iodine swabProduct Information Item Code (Source) NDC:67777-419 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength POVIDONE-IODINE (UNII: 85H0HZU99M) (IODINE - UNII:9679TC07X4) IODINE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength GLYCERIN (UNII: PDC6A3C0OX) POLYSORBATE 80 (UNII: 6OZP39ZG8H) SODIUM CITRATE (UNII: 1Q73Q2JULR) SODIUM PHOSPHATE, DIBASIC, ANHYDROUS (UNII: 22ADO53M6F) CITRIC ACID ACETATE (UNII: DSO12WL7AU) WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:67777-419-02 0.9 mL in 1 PACKET; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 09/13/2016 Part 4 of 4 ISOPROPYL ALCOHOL
isopropyl alcohol swabProduct Information Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength ISOPROPYL ALCOHOL (UNII: ND2M416302) (ISOPROPYL ALCOHOL - UNII:ND2M416302) ISOPROPYL ALCOHOL 70 mL in 100 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 5 mL in 1 POUCH; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC Monograph Drug M003 01/01/2007 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date unapproved drug other 07/25/2014 Labeler - Asclemed USA, Inc. (059888437)


