Label: METHYLENE BLUE INJ- methylene blue anhydrous injection, solution
- NDC Code(s): 42494-419-05
- Packager: Cameron Pharmaceuticals
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: unapproved drug other
DISCLAIMER: This drug has not been found by FDA to be safe and effective, and this labeling has not been approved by FDA. For further information about unapproved drugs, click here.
Drug Label Information
Updated August 9, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
-
BOXED WARNING
(What is this?)
WARNING: SEROTONIN SYNDROME WITH CONCOMITANT USE OF SEROTONERGIC DRUGS
Methylene Blue Injection may cause serious or fatal serotonergic syndrome when used in combination with serotonergic drugs. Avoid concomitant use of Methylene Blue Injection with selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), and monoamine oxidase inhibitors (see WARNINGS and PRECAUTIONS, Drug Interactions).
-
DESCRIPTION
Methylene Blue Injection is a sterile solution of Phenothiazin-5-ium, 3, 7-bis (dimethylamino)-, chloride, trihydrate. Each mL contains methylene blue, 10 mg in water for injection q.s. pH adjusted with hydrochloric acid and/or sodium hydroxide when necessary.
The structural formula is:
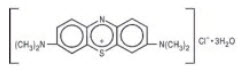
The molecular formula is:
C16H18ClN3S ∙ 3H2O MW = 373.90
-
CLINICAL PHARMACOLOGY
Methylene blue will produce two opposite actions on hemoglobin. Low concentrations will convert methemoglobin to hemoglobin. High concentrations convert the ferrous iron of reduced hemoglobin to ferric iron which results in the formation of methemoglobin.
Methylene blue is metabolized in the body to leukomethylene blue which is excreted primarily in the urine. Some unchanged drug is also excreted in the urine. (1)
- INDICATIONS AND USAGE
-
CONTRAINDICATIONS
Methylene blue can cause fetal harm when administered to a pregnant woman. An association exists between the use of methylene blue in amniocentesis and atresia of the ileum and jejunum, ileal occlusions, and other adverse effects in the neonate. (2, 3) Methylene blue is contraindicated in women who are or may become pregnant. If this drug is used during pregnancy, or if the patient becomes pregnant while taking this drug, the patient should be apprised of the potential hazard to the fetus.
Intraspinal and subcutaneous injections are contraindicated.
Methylene blue is contraindicated in patients with a known hypersensitivity to the drug.
-
WARNINGS
Methylene blue should not be given by subcutaneous or intrathecal injection.
Methylene blue is a potent monoamine oxidase inhibitor: Methylene blue has been demonstrated to be a potent monoamine oxidase inhibitor (MAOI) and may cause potentially fatal serotonin toxicity (serotonin syndrome) when combined with serotonin reuptake inhibitors (SRIs). (4) (See Drug Interactions.) Serotonin toxicity is characterized by development of neuromuscular hyperactivity (tremor, clonus, myoclonus and hyperreflexia, and, in the advanced stage, pyramidal rigidity); autonomic hyperactivity (diaphoresis, fever, tachycardia, tachypnoea, and mydriasis); and altered mental status (agitation, excitement, and in the advanced stage, confusion). If methylene blue is judged to be indicated, SRIs must be ceased, prior to treatment/procedure/surgery.
-
PRECAUTIONS
Drug Interactions
Methylene blue may interact with any drug that acts as a serotonin reuptake inhibitor (SRI) including, amongst others, selective serotonin reuptake inhibitors (SSRIs), serotonin and norepinephrine reuptake inhibitors (SNRIs), tricyclic antidepressants (TCAs), norepinephrine-dopamine reuptake inhibitors (NDRIs), triptans and ergot alkaloids; such combinations may have the consequence of potentially fatal serotonin toxicity (serotonin syndrome). Methylene blue should not be co-administered with any drug that acts as an SRI.
Pregnancy
Pregnancy Category X
Epidemiologic evidence exists that methylene blue is a teratogen. An association exists between the use of methylene blue in amniocentesis and atresia of the ileum and jejunum, ileal occlusions, and other adverse effects in the neonate. (2,3) Methylene blue injection should not be administered to pregnant women during amniocentesis due to the risk of teratogenicity and other newborn adverse effects (see CONTRAINDICATIONS).
Glucose-6-Phosphate Dehydrogenase Deficiency (G6PD Deficiency)
Methylene blue should be avoided in patients with G6PD deficiency due to the risk of paradoxical methemoglobinemia and hemolysis. (5,6)
Renal Failure
Methylene blue should be used with caution in patients with severe renal impairment (see CLINICAL PHARMACOLOGY).
Methylene blue must be injected intravenously very slowly over a period of several minutes to prevent local high concentration of the compound from producing additional methemoglobin. Do not exceed recommended dosage.
Large intravenous doses of methylene blue produce nausea, abdominal and precordial pain, dizziness, headache, profuse sweating, mental confusion and the formation of methemoglobin.
-
DOSAGE AND ADMINISTRATION
0.1 to 0.2 mL per kg body weight (0.045 to 0.09 mL per pound body weight). Inject methylene blue intravenously very slowly over a period of several minutes.
Methylene blue must be injected intravenously very slowly over a period of several minutes to prevent local high concentration of the compound from producing additional methemoglobin. Do not exceed recommended dosage.
Parenteral drug products should be inspected visually for particulate matter and discoloration, whenever solution and container permit.
To report SUSPECTED ADVERSE REACTIONS, contact: Cameron Pharmaceuticals, LLC at 1-888-296-9383 (or FDA 1-800-FDA-1088) or through email at www.fda.gov/medwatch.
-
HOW SUPPLIED
Methylene Blue Injection, 1%, USP is supplied as follows:
NDC 42494-419-05
10 mL vials in packages of 5.
The vials are packaged with a Flip Tear-Off Seal. The seal can either be flipped normally to reveal the rubber stopper or be totally removed so the rubber stopper can be taken out of the vial. The plastic button is attached to the metal seal, which when pulled, tears the seal at the score line allowing the metal portion to be removed.
- SPL UNCLASSIFIED SECTION
-
PRINCIPAL DISPLAY PANEL - 10 mg/mL Vial Carton Label
NDC 42494-419-05
Methylene Blue 1%
Injection, USP1% (10mg/mL)
For slow Intravenous administration
Rx only5 x 10ml Single Dose Vials
Each mL contains:
Methylene Blue 10 mg and Water
for Injection q.s. pH adjusted with
Hydrochloric Acid and/or Sodium
Hydroxide when necessary.Usual Dosage:
See package insert
for dosage informationManufactured for:
Cameron Pharmaceuticals, LLCEXP: 07/2023
LOT: 21VMBI024
-
INGREDIENTS AND APPEARANCE
METHYLENE BLUE INJ
methylene blue anhydrous injection, solutionProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:42494-419 Route of Administration INTRAVENOUS Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength METHYLENE BLUE ANHYDROUS (UNII: 8NAP7826UB) (METHYLENE BLUE CATION - UNII:ZMZ79891ZH) METHYLENE BLUE 10 mg in 1 mL Inactive Ingredients Ingredient Name Strength Water (UNII: 059QF0KO0R) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:42494-419-05 5 in 1 CARTON 08/07/2023 1 10 mL in 1 VIAL; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date Unapproved drug other 08/07/2023 Labeler - Cameron Pharmaceuticals (078371442) Establishment Name Address ID/FEI Business Operations Vitae Enim Vitae Scientific, Inc. 080492645 MANUFACTURE(42494-419)

