Label: PREZCOBIX- darunavir ethanolate and cobicistat tablet, film coated
- NDC Code(s): 59676-575-30, 59676-578-30
- Packager: Janssen Products LP
- Category: HUMAN PRESCRIPTION DRUG LABEL
- DEA Schedule: None
- Marketing Status: New Drug Application
Drug Label Information
Updated April 2, 2025
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
-
HIGHLIGHTS OF PRESCRIBING INFORMATION
These highlights do not include all the information needed to use PREZCOBIX safely and effectively. See full prescribing information for PREZCOBIX.
PREZCOBIX ®(darunavir and cobicistat) tablets, for oral use
Initial U.S. Approval: 2015INDICATIONS AND USAGE
PREZCOBIX is a two-drug combination of darunavir, a human immunodeficiency virus (HIV-1) protease inhibitor, and cobicistat, a CYP3A inhibitor, and is indicated for the treatment of HIV-1 in treatment-naïve and treatment-experienced adults and pediatric patients weighing at least 25 kg with no darunavir resistance-associated substitutions (V11I, V32I, L33F, I47V, I50V, I54L, I54M, T74P, L76V, I84V, L89V). ( 1)
DOSAGE AND ADMINISTRATION
Recommended dosage:
- Adults and pediatric patients weighing at least 40 kg: One 800 mg/150 mg tablet taken once daily with food. ( 2.1)
- Pediatric patients weighing at least 25 kg to less than 40 kg: One 675 mg/150 mg tablet taken once daily with food. ( 2.1)
Testing Prior to Initiation:HIV genotypic testing is recommended for antiretroviral treatment experienced patients. Assess estimated creatinine clearance in all patients prior to starting PREZCOBIX. When used with tenofovir DF: Assess urine glucose and urine protein at baseline and monitor creatinine clearance, urine glucose, and urine protein. Monitor serum phosphorus in patients with or at risk for renal impairment. ( 2.2)
DOSAGE FORMS AND STRENGTHS
CONTRAINDICATIONS
WARNINGS AND PRECAUTIONS
- Drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis), liver injury, including some fatalities can occur with PREZCOBIX. Monitor liver function before and during therapy, especially in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases. ( 5.1)
- Skin reactions ranging from mild to severe, including Stevens-Johnson Syndrome, toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms and acute generalized exanthematous pustulosis, can occur with PREZCOBIX. Discontinue treatment if severe reaction develops. ( 5.2)
- When PREZCOBIX is used in combination with a tenofovir disoproxil fumarate (tenofovir DF) containing regimen, cases of acute renal failure and Fanconi syndrome have been reported. ( 5.4)
- PREZCOBIX is not recommended in combination with other antiretroviral drugs that require pharmacokinetic boosting. ( 5.6)
- Monitor in patients with a known sulfonamide allergy. ( 5.7)
- Patients receiving PREZCOBIX may develop new onset or exacerbations of diabetes mellitus/hyperglycemia ( 5.8), redistribution/accumulation of body fat ( 5.9), and immune reconstitution syndrome. ( 5.10)
- Patients with hemophilia may develop increased bleeding events. ( 5.11)
ADVERSE REACTIONS
- The most common adverse reactions to darunavir, a component of PREZCOBIX (incidence greater than or equal to 5%) of at least moderate severity (greater than or equal to Grade 2) were diarrhea, nausea, rash, headache, abdominal pain, and vomiting. ( 6.1)
To report SUSPECTED ADVERSE REACTIONS, contact Janssen Products, LP at 1-800-JANSSEN (1-800-526-7736) or FDA at 1-800-FDA-1088 or www.fda.gov/medwatch.
DRUG INTERACTIONS
USE IN SPECIFIC POPULATIONS
See 17 for PATIENT COUNSELING INFORMATION and FDA-approved patient labeling.
Revised: 3/2025
-
Table of Contents
FULL PRESCRIBING INFORMATION: CONTENTS*
1 INDICATIONS AND USAGE
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage in Adults and Pediatric Patients Weighing at Least 25 kg
2.2 Testing Prior to Initiation of PREZCOBIX
2.3 Not Recommended in Severe Renal Impairment
2.4 Not Recommended in Severe Hepatic Impairment
2.5 Not Recommended During Pregnancy
3 DOSAGE FORMS AND STRENGTHS
4 CONTRAINDICATIONS
5 WARNINGS AND PRECAUTIONS
5.1 Hepatotoxicity
5.2 Severe Skin Reactions
5.3 Effects on Serum Creatinine
5.4 New Onset or Worsening Renal Impairment When Used With Tenofovir Disoproxil Fumarate
5.5 Risk of Serious Adverse Reactions or Loss of Virologic Response Due to Drug Interactions
5.6 Antiretrovirals Not Recommended
5.7 Sulfa Allergy
5.8 Diabetes Mellitus/Hyperglycemia
5.9 Fat Redistribution
5.10 Immune Reconstitution Syndrome
5.11 Hemophilia
6 ADVERSE REACTIONS
6.1 Clinical Trials Experience
6.2 Postmarketing Experience
7 DRUG INTERACTIONS
7.1 Potential for PREZCOBIX to Affect Other Drugs
7.2 Potential for Other Drugs to Affect PREZCOBIX
7.3 Established and Other Potentially Significant Drug Interactions
7.4 Drugs without Clinically Significant Interactions with PREZCOBIX
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
8.2 Lactation
8.3 Females and Males of Reproductive Potential
8.4 Pediatric Use
8.5 Geriatric Use
8.6 Hepatic Impairment
8.7 Renal Impairment
10 OVERDOSAGE
11 DESCRIPTION
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
12.2 Pharmacodynamics
12.3 Pharmacokinetics
12.4 Microbiology
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
14 CLINICAL STUDIES
14.1 Clinical Trial Results in Adults with HIV-1
14.2 Clinical Trial Results in Pediatrics with HIV-1
16 HOW SUPPLIED/STORAGE AND HANDLING
17 PATIENT COUNSELING INFORMATION
- *
- Sections or subsections omitted from the full prescribing information are not listed.
-
1 INDICATIONS AND USAGE
PREZCOBIX is indicated in combination with other antiretroviral agents for the treatment of human immunodeficiency virus (HIV-1) in treatment-naïve and treatment-experienced adults and pediatric patients weighing at least 25 kg with no darunavir resistance-associated substitutions (V11I, V32I, L33F, I47V, I50V, I54L, I54M, T74P, L76V, I84V, L89V).
-
2 DOSAGE AND ADMINISTRATION
2.1 Recommended Dosage in Adults and Pediatric Patients Weighing at Least 25 kg
The recommended dosages of PREZCOBIX for adults and pediatric patients weighing at least 25 kg are shown in Table 1. The pediatric dose is based on weight. Administer PREZCOBIX orally with food in conjunction with other antiretroviral agents.
Table 1: Recommended Dosages of PREZCOBIX in Adults and Pediatric Patients Weighing at Least 25 kg, who are Treatment-Naïve or Treatment-Experienced Without DRV RAMs * Patient Population Dose (once daily with food) - *
- DRV-resistance-associated mutations (RAMs): V11I, V32I, L33F, I47V, I50V, I54M, I54L, T74P, L76V, I84V, L89V.
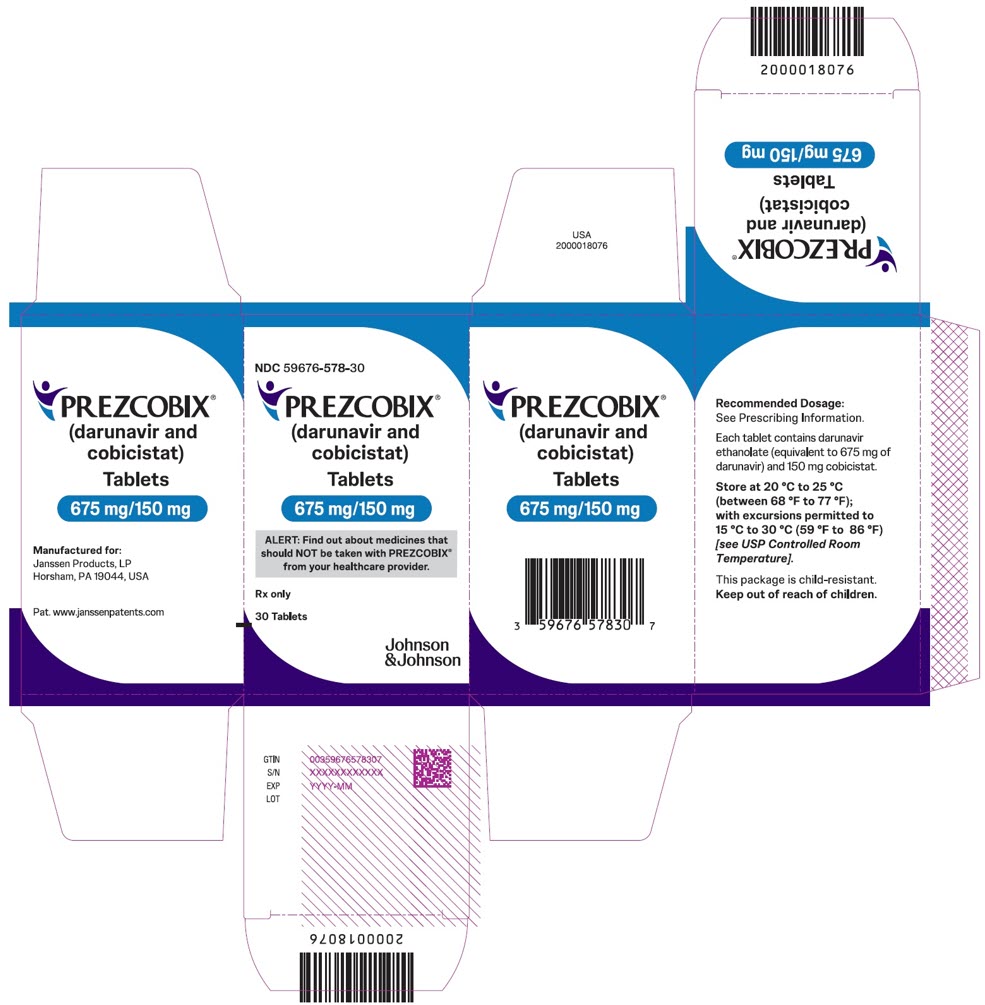
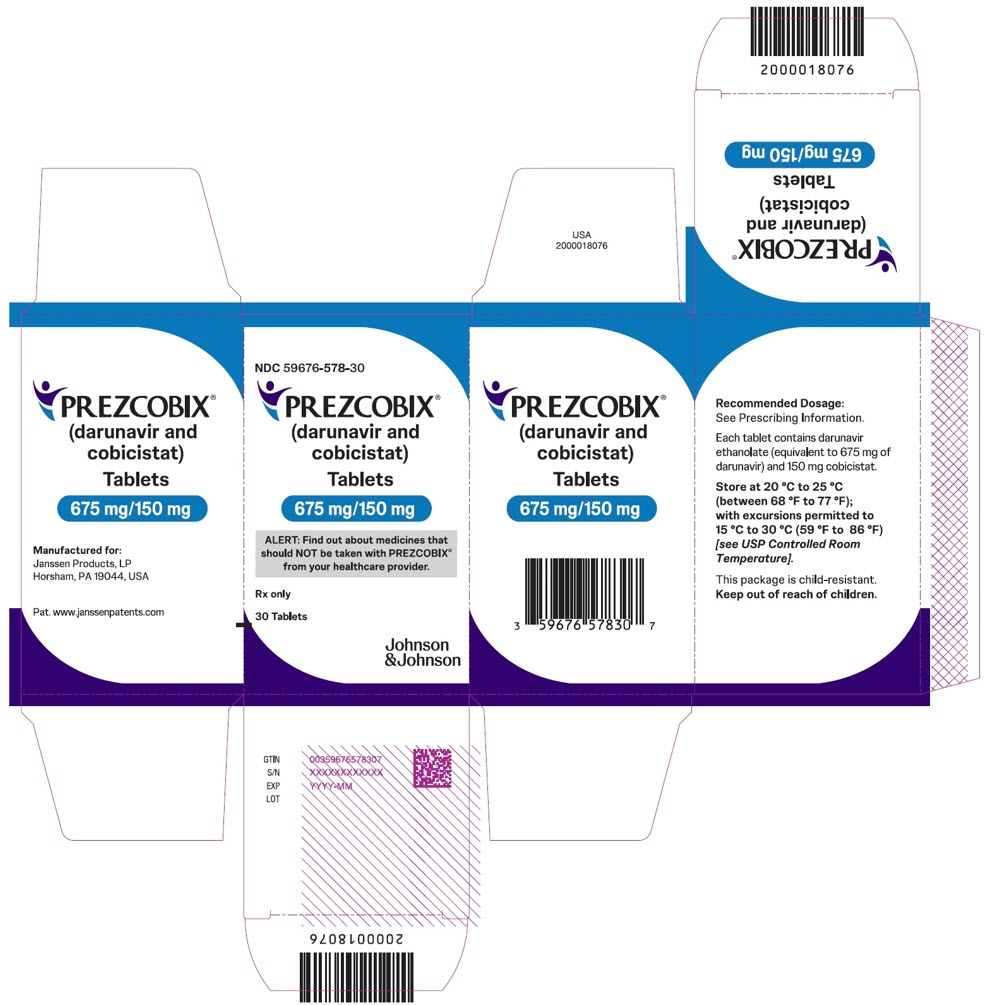
Adult Patients One 800 mg darunavir/150 mg cobicistat tablet Pediatric Patients weighing at least 40 kg Pediatric Patients weighing at least 25 kg to less than 40 kg One 675 mg darunavir/150 mg cobicistat tablet Before prescribing PREZCOBIX 675 mg/150 mg tablets, children should be assessed for the ability to swallow tablets. For patients unable to swallow the 675 mg/150 mg tablet whole, the scored tablet may be split by hand into two pieces. Each piece should be consumed immediately after splitting to ensure the entire dose is administered. The score line is only to facilitate breaking for ease of swallowing and not to divide into equal doses.
2.2 Testing Prior to Initiation of PREZCOBIX
HIV Genotypic Testing
HIV genotypic testing is recommended for antiretroviral treatment-experienced patients. However, when HIV genotypic testing is not feasible, PREZCOBIX can be used in protease inhibitor-naïve patients, but is not recommended in protease inhibitor-experienced patients.
Creatinine Clearance
Prior to starting PREZCOBIX, assess estimated creatinine clearance because cobicistat decreases estimated creatinine clearance due to inhibition of tubular secretion of creatinine without affecting actual renal glomerular function [see Warnings and Precautions (5.3)] . When co-administering PREZCOBIX with tenofovir disoproxil fumarate (tenofovir DF) assess estimated creatinine clearance, urine glucose, and urine protein at baseline [see Warnings and Precautions (5.4)] .
2.3 Not Recommended in Severe Renal Impairment
PREZCOBIX co-administered with tenofovir DF is not recommended in patients who have an estimated creatinine clearance below 70 mL per minute [see Warnings and Precautions (5.4)and Adverse Reactions (6.1)] .
2.4 Not Recommended in Severe Hepatic Impairment
PREZCOBIX is not recommended for use in patients with severe hepatic impairment [see Use in Specific Populations (8.6)and Clinical Pharmacology (12.3)] .
2.5 Not Recommended During Pregnancy
PREZCOBIX is not recommended during pregnancy because of substantially lower exposures of darunavir and cobicistat during the second and third trimesters [see Use in Specific Populations (8.1)and Clinical Pharmacology (12.3)] .
PREZCOBIX should not be initiated in pregnant individuals. An alternative regimen is recommended for those who become pregnant during therapy with PREZCOBIX.
-
3 DOSAGE FORMS AND STRENGTHS
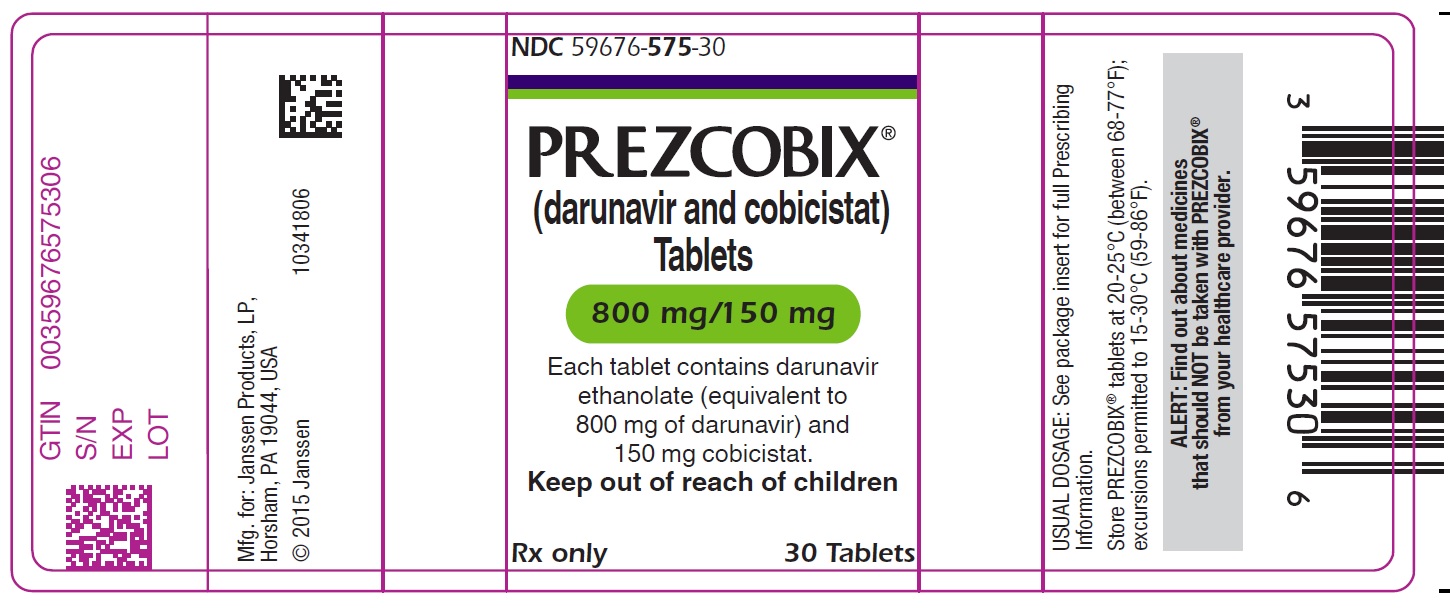
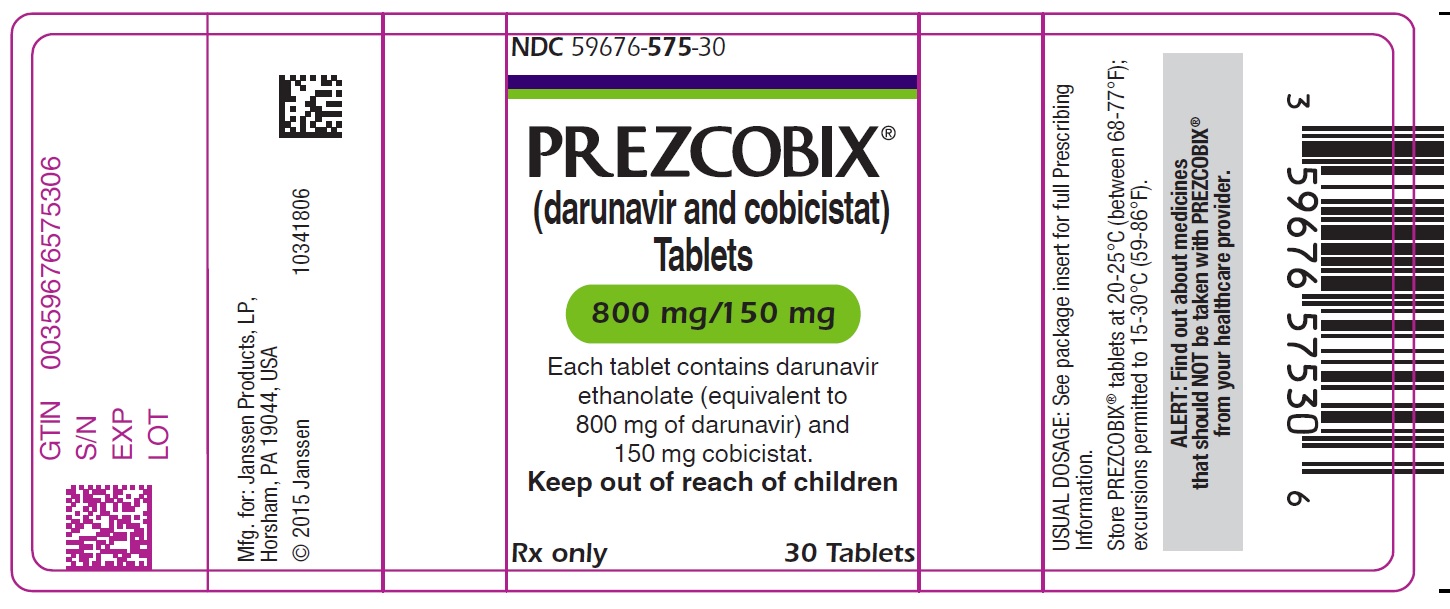
PREZCOBIX 800 mg darunavir/150 mg cobicistat is a pink, oval-shaped, film-coated tablet debossed with “800” on one side and “TG” on the other side.
PREZCOBIX 675 mg darunavir/150 mg cobicistat is a green to dark green, oval-shaped, scored film-coated tablet debossed with “675” on one side and “TG” on the other side.
-
4 CONTRAINDICATIONS
Darunavir and cobicistat are both inhibitors of the cytochrome P450 3A (CYP3A) isoform. PREZCOBIX should not be co-administered with medicinal products that are highly dependent on CYP3A for clearance and for which increased plasma concentrations are associated with serious and/or life-threatening events (narrow therapeutic index). Darunavir and cobicistat are both substrates of the cytochrome P450 3A (CYP3A) isoform. Co-administration of PREZCOBIX with CYP3A inducers may lead to lower exposures of darunavir and cobicistat and potential loss of efficacy of darunavir and possible resistance. Examples of drugs that are contraindicated for co-administration with PREZCOBIX [see Drug Interactions (7.3)and Clinical Pharmacology (12.3)] are listed below.
- Alpha 1-adrenoreceptor antagonist: alfuzosin
- Anticonvulsants: carbamazepine, phenobarbital, phenytoin
- Anti-gout: colchicine, in patients with renal and/or hepatic impairment
- Antimycobacterial: rifampin
- Antipsychotics: lurasidone, pimozide
- Cardiac Disorders: dronedarone, ivabradine, ranolazine
- Ergot derivatives, e.g. dihydroergotamine, ergotamine, methylergonovine
- Herbal product: St. John's wort ( Hypericum perforatum)
- Hepatitis C direct acting antiviral: elbasvir/grazoprevir
- Lipid modifying agents: lomitapide, lovastatin, simvastatin
- Opioid Antagonist: naloxegol
- PDE-5 inhibitor: sildenafil when used for treatment of pulmonary arterial hypertension
- Sedatives/hypnotics: orally administered midazolam, triazolam
-
5 WARNINGS AND PRECAUTIONS
5.1 Hepatotoxicity
During the darunavir clinical development program (N=3063), where darunavir was co-administered with ritonavir 100 mg once or twice daily, drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis) was reported in 0.5% of participants. Patients with pre-existing liver dysfunction, including chronic active hepatitis B or C, have an increased risk for liver function abnormalities including severe hepatic adverse reactions.
Post-marketing cases of liver injury, including some fatalities, have also been reported with darunavir co-administered with ritonavir. These have generally occurred in patients with advanced HIV-1 disease taking multiple concomitant medications, having co-morbidities including hepatitis B or C co-infection, and/or developing immune reconstitution syndrome. A causal relationship with darunavir co-administered with ritonavir has not been established.
Appropriate laboratory testing should be conducted prior to initiating therapy with PREZCOBIX and patients should be monitored during treatment. Increased AST/ALT monitoring should be considered in patients with underlying chronic hepatitis, cirrhosis, or in patients who have pre-treatment elevations of transaminases, especially during the first several months of PREZCOBIX treatment.
Evidence of new or worsening liver dysfunction (including clinically significant elevation of liver enzymes and/or symptoms such as fatigue, anorexia, nausea, jaundice, dark urine, liver tenderness, hepatomegaly) in patients on PREZCOBIX should prompt consideration of interruption or discontinuation of treatment.
5.2 Severe Skin Reactions
During the darunavir clinical development program (n=3063), where darunavir was co-administered with ritonavir 100 mg once or twice daily, severe skin reactions, accompanied by fever and/or elevations of transaminases in some cases, was reported in 0.4% of participants. Stevens-Johnson Syndrome was rarely (less than 0.1%) reported during the clinical development program. During post-marketing experience toxic epidermal necrolysis, drug rash with eosinophilia and systemic symptoms, and acute generalized exanthematous pustulosis have been reported. Discontinue PREZCOBIX immediately if signs or symptoms of severe skin reactions develop. These can include but are not limited to severe rash or rash accompanied with fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, conjunctivitis, hepatitis and/or eosinophilia.
Mild-to-moderate rash was also reported and often occurred within the first four weeks of treatment and resolved with continued dosing.
5.3 Effects on Serum Creatinine
Cobicistat decreases estimated creatinine clearance due to inhibition of tubular secretion of creatinine without affecting actual renal glomerular function. This effect should be considered when interpreting changes in estimated creatinine clearance in patients initiating PREZCOBIX, particularly in patients with medical conditions or receiving drugs needing monitoring with estimated creatinine clearance.
Prior to initiating therapy with PREZCOBIX, assess estimated creatinine clearance [see Dosage and Administration (2.2)] . Dosage recommendations are not available for drugs that require dosage adjustments in PREZCOBIX-treated patients with renal impairment [see Drug Interactions (7.3)and Clinical Pharmacology (12.2)] . Consider alternative medications that do not require dosage adjustments in patients with renal impairment.
Although cobicistat may cause modest increases in serum creatinine and modest declines in estimated creatinine clearance without affecting renal glomerular function, patients who experience a confirmed increase in serum creatinine of greater than 0.4 mg/dL from baseline should be closely monitored for renal safety.
5.4 New Onset or Worsening Renal Impairment When Used With Tenofovir Disoproxil Fumarate
Renal impairment, including cases of acute renal failure and Fanconi syndrome, has been reported when cobicistat, a component of PREZCOBIX, was used in an antiretroviral regimen that contained tenofovir DF. Co-administration of PREZCOBIX and tenofovir DF is not recommended in patients who have an estimated creatinine clearance below 70 mL/min [see Dosage and Administration (2.3)] .
- Document urine glucose and urine protein at baseline [see Dosage and Administration (2.2)] and perform routine monitoring of estimated creatinine clearance, urine glucose, and urine protein during treatment when PREZCOBIX is used with tenofovir DF. Measure serum phosphorus in patients with or at risk for renal impairment when used with tenofovir DF.
- Co-administration of PREZCOBIX and tenofovir DF in combination with concomitant or recent use of a nephrotoxic agent is not recommended.
See cobicistat full prescribing information for additional information regarding cobicistat.
5.5 Risk of Serious Adverse Reactions or Loss of Virologic Response Due to Drug Interactions
Initiation of PREZCOBIX, which inhibits CYP3A, in patients receiving medications metabolized by CYP3A, or initiation of medications metabolized by CYP3A in patients already receiving PREZCOBIX may increase plasma concentrations of medications metabolized by CYP3A and reduce plasma concentrations of active metabolite(s) formed by CYP3A. Initiation of medications that inhibit or induce CYP3A may respectively increase or decrease concentrations of PREZCOBIX.
These interactions may lead to:
- clinically significant adverse reactions, potentially leading to severe, life threatening, or fatal events from higher exposures of concomitant medications.
- clinically significant adverse reactions from higher exposures of PREZCOBIX.
- loss of therapeutic effect of the concomitant medications from lower exposures of active metabolite(s).
- loss of therapeutic effect of PREZCOBIX and possible development of resistance from lower exposures of PREZCOBIX.
See Table 2 for steps to prevent or manage these possible and known significant drug interactions, including dosing recommendations. Consider the potential for drug interactions prior to and during PREZCOBIX therapy; review concomitant medications during PREZCOBIX therapy; and monitor for the adverse reactions associated with concomitant medications [see Contraindications (4) and Drug Interactions (7)] .
When used with concomitant medications, PREZCOBIX may result in different drug interactions than those observed or expected with darunavir co-administered with ritonavir. Complex or unknown mechanisms of drug interactions preclude extrapolation of drug interactions with darunavir co-administered with ritonavir to certain PREZCOBIX interactions [see Drug Interactions (7) and Clinical Pharmacology (12.3)] .
5.6 Antiretrovirals Not Recommended
PREZCOBIX is not recommended in combination with other antiretroviral drugs that require pharmacokinetic boosting (i.e., another protease inhibitor or elvitegravir) because dosing recommendations for such combinations have not been established and co-administration may result in decreased plasma concentrations of the antiretroviral agents, leading to loss of therapeutic effect and development of resistance.
PREZCOBIX is not recommended in combination with products containing the individual components of PREZCOBIX (darunavir and cobicistat) or with ritonavir. For additional recommendations on use of PREZCOBIX with other antiretroviral agents, [see Drug Interactions (7)] .
5.7 Sulfa Allergy
Darunavir contains a sulfonamide moiety. Monitor patients with a known sulfonamide allergy after initiating PREZCOBIX. In clinical studies with darunavir co-administered with ritonavir, the incidence and severity of rash were similar in subjects with or without a history of sulfonamide allergy.
5.8 Diabetes Mellitus/Hyperglycemia
New onset diabetes mellitus, exacerbation of pre-existing diabetes mellitus, and hyperglycemia have been reported during postmarketing surveillance in patients with HIV-1 receiving HIV protease inhibitor (PI) therapy. Some patients required either initiation or dose adjustments of insulin or oral hypoglycemic agents for treatment of these events. In some cases, diabetic ketoacidosis has occurred. In those patients who discontinued PI therapy, hyperglycemia persisted in some cases. Because these events have been reported voluntarily during clinical practice, estimates of frequency cannot be made and causal relationships between HIV PI therapy and these events have not been established.
5.9 Fat Redistribution
Redistribution/accumulation of body fat, including central obesity, dorsocervical fat enlargement (buffalo hump), peripheral wasting, facial wasting, breast enlargement, and "cushingoid appearance" have been observed in patients receiving antiretroviral therapy. The mechanism and long-term consequences of these events are currently unknown. A causal relationship has not been established.
5.10 Immune Reconstitution Syndrome
Immune reconstitution syndrome has been reported in patients treated with combination antiretroviral therapy, including PREZCOBIX. During the initial phase of combination antiretroviral treatment, patients whose immune systems respond may develop an inflammatory response to indolent or residual opportunistic infections (such as Mycobacterium avium infection, cytomegalovirus, Pneumocystis jirovecii pneumonia [PCP], or tuberculosis), which may necessitate further evaluation and treatment.
Autoimmune disorders (such as Graves' disease, polymyositis, Guillain-Barré syndrome and autoimmune hepatitis) have also been reported to occur in the setting of immune reconstitution; however, the time to onset is more variable, and can occur many months after initiation of antiretroviral treatment.
5.11 Hemophilia
There have been reports of increased bleeding, including spontaneous skin hematomas and hemarthrosis in patients with hemophilia type A and B treated with HIV PIs. In some patients, additional factor VIII was given. In more than half of the reported cases, treatment with HIV PIs was continued or reintroduced if treatment had been discontinued. A causal relationship between PI therapy and these episodes has not been established.
-
6 ADVERSE REACTIONS
The following adverse reactions are discussed in other sections of the labeling:
- Hepatotoxicity [see Warnings and Precautions (5.1)]
- Severe skin reactions [see Warnings and Precautions (5.2)]
- Effects on serum creatinine [see Warnings and Precautions (5.3)]
- New onset or worsening renal impairment when used with tenofovir DF [see Warnings and Precautions (5.4)]
- Immune Reconstitution Syndrome [see Warnings and Precautions (5.10)]
6.1 Clinical Trials Experience
Because clinical trials are conducted under widely varying conditions, adverse reaction rates observed in the clinical trials of a drug cannot be directly compared to rates in the clinical trials of another drug and may not reflect the rates observed in clinical practice.
Clinical Trials in Adults
During the darunavir clinical development program, where darunavir was co-administered with ritonavir 100 mg once or twice daily, the most common clinical adverse reactions (incidence greater than or equal to 5%) of at least moderate intensity (greater than or equal to Grade 2) were diarrhea, nausea, rash, headache, abdominal pain, and vomiting. See the darunavir full prescribing information for additional information on adverse reactions reported with darunavir co-administered with ritonavir. See cobicistat full prescribing information for clinical trial information on adverse reactions reported with cobicistat.
One single arm clinical trial was conducted with darunavir and cobicistat administered as single entities in 313 participants with HIV-1. Adverse reactions evaluated through Week 24 did not differ substantially from those reported in clinical trials with darunavir co-administered with ritonavir.
Clinical Trials in Pediatrics
No clinical trials with PREZCOBIX were performed in pediatric patients. However, the safety of the components of PREZCOBIX, darunavir and cobicistat, co-administered with two nucleoside reverse transcriptase inhibitors, was evaluated in pediatric participants of 12 to less than 18 years of age with HIV-1 through clinical trial GS-US-216-0128 (Cohort 1, virologically-suppressed, N=7 with weight ≥40 kg) and pediatric participants 6 to less than 12 years of age (Cohort 2, virologically-suppressed, N=8 with weight ≥25 kg) through Week 48. Safety analyses of this trial in these pediatric participants did not identify new safety concerns compared to the known safety profile of PREZCOBIX in adult participants [see Clinical Studies (14.2)] .
6.2 Postmarketing Experience
The following adverse reactions have been identified during post-approval use of darunavir. Because these reactions are reported voluntarily from a population of uncertain size, it is not always possible to reliably estimate their frequency or establish a causal relationship to drug exposure.
Musculoskeletal and Connective Tissue Disorders
Rhabdomyolysis (associated with co-administration with HMG-CoA reductase inhibitors)
Skin and Subcutaneous Tissue Disorders
Toxic epidermal necrolysis, acute generalized exanthematous pustulosis, drug rash with eosinophilia and systemic symptoms [see Warnings and Precautions (5.2)] .
-
7 DRUG INTERACTIONS
7.1 Potential for PREZCOBIX to Affect Other Drugs
Darunavir co-administered with cobicistat is an inhibitor of CYP3A and CYP2D6. Cobicistat inhibits the following transporters: P-glycoprotein (P-gp), BCRP, MATE1, OATP1B1 and OATP1B3. Therefore, co-administration of PREZCOBIX with drugs that are primarily metabolized by CYP3A and/or CYP2D6 or are substrates of P-gp, BCRP, MATE1, OATP1B1 or OATP1B3 may result in increased plasma concentrations of such drugs, which could increase or prolong their therapeutic effect and can be associated with adverse events. Co-administration of PREZCOBIX with drugs that have active metabolite(s) formed by CYP3A may result in reduced plasma concentrations of these active metabolite(s), potentially leading to loss of their therapeutic effect (see Table 2).
7.2 Potential for Other Drugs to Affect PREZCOBIX
Darunavir is metabolized by CYP3A. Cobicistat is metabolized by CYP3A, and to a minor extent, by CYP2D6. Co-administration of PREZCOBIX and drugs that induce CYP3A activity are expected to increase the clearance of darunavir and cobicistat, resulting in lowered plasma concentrations of darunavir and cobicistat which may lead to loss of therapeutic effect and development of resistance. Co-administration of PREZCOBIX and other drugs that inhibit CYP3A may result in increased plasma concentrations of darunavir and cobicistat (see Table 2).
7.3 Established and Other Potentially Significant Drug Interactions
Table 2 provides dosing recommendations for expected clinically relevant interactions with PREZCOBIX (this table is not all inclusive). These recommendations are based on either drug interaction trials or predicted interactions due to the expected magnitude of interaction and potential for serious adverse events or loss of therapeutic effect. The table includes examples of potentially significant interactions but is not all inclusive , and therefore the label of each drug that is co-administered with PREZCOBIX should be consulted for information related to the route of metabolism, interaction pathways, potential risks, and specific actions to be taken with regard to co-administration. For the list of examples of contraindicated drugs, [see Contraindications (4)] .
Table 2: Established and Other Potentially Significant *Drug Interactions: Alterations in Dose or Regimen May Be Recommended Concomitant Drug Class:
Drug Name ExamplesEffect on Concentration of Darunavir, Cobicistat, or Concomitant Drug Clinical Comment - *
- this table is not all inclusive
HIV-1 antiviral agents: Nucleoside Reverse Transcriptase Inhibitors (NRTIs) didanosine ↔ darunavir
↔ cobicistat
↔ didanosineDidanosine should be administered one hour before or two hours after PREZCOBIX (administered with food). HIV-1 antiviral agents: Non-Nucleoside Reverse Transcriptase Inhibitors (NNRTIs) efavirenz ↓ cobicistat
↓ darunavirCo-administration with efavirenz is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. etravirine ↓ cobicistat
darunavir: effect unknownCo-administration with etravirine is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. nevirapine ↓ cobicistat
darunavir: effect unknownCo-administration with nevirapine is not recommended because it may result in loss of therapeutic effect and development of resistance to darunavir. HIV-1 antiviral agents: CCR5 co-receptor antagonists maraviroc ↑ maraviroc Maraviroc is a substrate of CYP3A. When co-administered with PREZCOBIX, patients should receive maraviroc 150 mg twice daily. Other agents Alpha 1-adrenoreceptor antagonist:
alfuzosin↑ alfuzosin Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as hypotension. Antibacterials:
clarithromycin, erythromycin, telithromycin↑ darunavir
↑ cobicistat
↑ antibacterialConsider alternative antibiotics with concomitant use of PREZCOBIX. Anticancer agents:
dasatinib, nilotinib↑ anticancer agent A decrease in the dosage or an adjustment of the dosing interval of dasatinib or nilotinib may be necessary when co-administered with PREZCOBIX. Consult the dasatinib and nilotinib prescribing information for dosing instructions. vinblastine, vincristine For vincristine and vinblastine, consider temporarily withholding the cobicistat-containing antiretroviral regimen in patients who develop significant hematologic or gastrointestinal side effects when PREZCOBIX is administered concurrently with vincristine or vinblastine. If the antiretroviral regimen must be withheld for a prolonged period, consider initiating a revised regimen that does not include a CYP3A or P-gp inhibitor. Anticoagulants:
Direct Oral Anticoagulants (DOACs)
apixaban↑ apixaban Due to potentially increased bleeding risk, dosing recommendations for co-administration of apixaban with PREZCOBIX depend on the apixaban dose. Refer to apixaban dosing instructions for co-administration with P-gp and strong CYP3A inhibitors in apixaban prescribing information. rivaroxaban ↑ rivaroxaban Co-administration of rivaroxaban with PREZCOBIX is not recommended because it may lead to an increased bleeding risk. dabigatran etexilate
edoxaban↑ dabigatran
↑ edoxabanRefer to the dabigatran etexilate or edoxaban prescribing information for recommendations regarding co-administration. The specific recommendations are based on indication, renal function, and effect of the co-administered P-gp inhibitors on the concentration of dabigatran or edoxaban. Clinical monitoring is recommended when a DOAC not affected by CYP3A4 but transported by P-gp, including dabigatran etexilate and edoxaban, is co-administered with PREZCOBIX. Other Anticoagulants: warfarin warfarin: effect unknown Monitor the international normalized ratio (INR) when co-administering with warfarin. Anticonvulsants:
carbamazepine, phenobarbital, phenytoin↓ darunavir
↓ cobicistatCo-administration is contraindicated due to potential for reduced plasma concentrations of darunavir, which may result in loss of therapeutic effect and development of resistance. Anticonvulsants with CYP3A induction effects that are NOT contraindicated:
e.g. eslicarbazepine, oxcarbazepine↓ cobicistat
darunavir: effect unknownConsider alternative anticonvulsant or antiretroviral therapy to avoid potential changes in exposures. If co-administration is necessary, monitor for lack or loss of virologic response. Anticonvulsants that are metabolized by CYP3A:
e.g. clonazepam↑ clonazepam Clinical monitoring of anticonvulsants is recommended. Antidepressants:
Selective Serotonin Reuptake Inhibitors (SSRIs):
e.g. paroxetine, sertralineSSRIs: effects unknown When co-administering with SSRIs, TCAs, or trazodone, careful dose titration of the antidepressant to the desired effect, including using the lowest feasible initial or maintenance dose, and monitoring for antidepressant response are recommended. Tricyclic Antidepressants (TCAs):
e.g. amitriptyline, desipramine, imipramine, nortriptyline↑ TCAs Other antidepressants:
trazodone↑ trazodone Antifungals:
itraconazole, isavuconazole, ketoconazole, posaconazole↑ darunavir
↑ cobicistatMonitor for increased darunavir or cobicistat and/or antifungal adverse reactions. ↑ itraconazole
↑ ketoconazole
↑ isavuconazole
↔ posaconazoleSpecific dosing recommendations are not available for co-administration with these antifungals. Monitor for increased itraconazole or ketoconazole adverse reactions. voriconazole voriconazole: effects unknown Co-administration with voriconazole is not recommended unless benefit/risk assessment justifies the use of voriconazole. Anti-gout:
colchicine↑ colchicine Co-administration is contraindicated in patients with renal and/or hepatic impairment due to potential for serious and/or life-threatening reactions.
For patients without renal or hepatic impairment:- Treatment of gout flares – co-administration of colchicine: 0.6 mg (1 tablet) × 1 dose, followed by 0.3 mg (half tablet) 1 hour later. Treatment course to be repeated no earlier than 3 days.
- Prophylaxis of gout flares – co-administration of colchicine: If the original regimen was 0.6 mg twice a day, the regimen should be adjusted to 0.3 mg once a day. If the original regimen was 0.6 mg once a day, the regimen should be adjusted to 0.3 mg once every other day.
- Treatment of familial Mediterranean fever – co-administration of colchicine: Maximum daily dose of 0.6 mg (may be given as 0.3 mg twice a day).
Antimalarial:
artemether/lumefantrineartemether: effect unknown
lumefantrine: effect unknownMonitor for a potential decrease of antimalarial efficacy or potential QT prolongation. Antimycobacterials: rifampin ↓ darunavir
↓ cobicistatCo-administration is contraindicated due to potential for loss of therapeutic effect and development of resistance. rifabutin ↑ rifabutin
cobicistat: effects unknown
darunavir: effects unknownWhen used in combination with PREZCOBIX, the recommended dose of rifabutin is 150 mg every other day. Monitor for rifabutin-associated adverse reactions including neutropenia and uveitis. rifapentine ↓ darunavir Co-administration with rifapentine is not recommended. Antipsychotics:
lurasidone↑ lurasidone Co-administration is contraindicated due to potential for serious and/or life-threatening reactions. pimozide ↑ pimozide Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. e.g. perphenazine, risperidone, thioridazine ↑ antipsychotic A decrease in the dose of antipsychotics that are metabolized by CYP3A or CYP2D6 may be needed when co-administered with PREZCOBIX. quetiapine ↑ quetiapine Initiation of PREZCOBIX in patients taking quetiapine: Consider alternative antiretroviral therapy to avoid increases in quetiapine exposure. If co-administration is necessary, reduce the quetiapine dose to 1/6 of the current dose and monitor for quetiapine- associated adverse reactions. Refer to the quetiapine prescribing information for recommendations on adverse reaction monitoring.
Initiation of quetiapine in patients taking PREZCOBIX: Refer to the quetiapine prescribing information for initial dosing and titration of quetiapine.β-Blockers:
e.g. carvedilol, metoprolol, timolol↑ beta-blockers Clinical monitoring is recommended for co-administration with beta-blockers that are metabolized by CYP2D6. Calcium channel blockers:
e.g. amlodipine, diltiazem, felodipine, nifedipine, verapamil↑ calcium channel blockers Clinical monitoring is recommended for co-administration with calcium channel blockers metabolized by CYP3A. Cardiac Disorders: ranolazine, ivabradine ↑ ranolazine
↑ ivabradineCo-administration is contraindicated due to potential for serious and/or life-threatening reactions. dronedarone ↑ dronedarone Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as cardiac arrhythmias. Other antiarrhythmics e.g. amiodarone, disopyramide, flecainide, lidocaine (systemic), mexiletine, propafenone, quinidine ↑ antiarrhythmics Clinical monitoring is recommended upon co-administration with antiarrhythmics. digoxin ↑ digoxin When co-administering with digoxin, titrate the digoxin dose and monitor digoxin concentrations. Corticosteroids:
dexamethasone (systemic)
Corticosteroids primarily metabolized by CYP3A:
e.g.
betamethasone
budesonide
ciclesonide
fluticasone
methylprednisolone
mometasone
triamcinolone↓ darunavir
↓ cobicistat
↑ corticosteroidsCo-administration with systemic dexamethasone or other systemic corticosteroids that induce CYP3A may result in loss of therapeutic effect and development of resistance to PREZCOBIX. Consider alternative corticosteroids.
Co-administration with corticosteroids (all routes of administration) of which exposures are significantly increased by strong CYP3A inhibitors can increase the risk for Cushing's syndrome and adrenal suppression.
Alternative corticosteroids including beclomethasone, prednisone and prednisolone (for which PK and/or PD are less affected by strong CYP3A inhibitors relative to other steroids) should be considered, particularly for long-term use.Endothelin receptor antagonists:
bosentan↓ darunavir
↓ cobicistat
↑ bosentanInitiation of bosentan in patients taking PREZCOBIX: In patients who have been receiving PREZCOBIX for at least 10 days, start bosentan at 62.5 mg once daily or every other day based upon individual tolerability.
Initiation of PREZCOBIX in patients on bosentan: Discontinue use of bosentan at least 36 hours prior to initiation of PREZCOBIX. After at least 10 days following the initiation of PREZCOBIX, resume bosentan at 62.5 mg once daily or every other day based upon individual tolerability.
Switching from darunavir co-administered with ritonavir to PREZCOBIX in patients on bosentan: Maintain bosentan dose.Ergot derivatives:
e.g. dihydroergotamine, ergotamine, methylergonovine↑ ergot derivatives Co-administration is contraindicated due to potential for serious and/or life-threatening reactions such as acute ergot toxicity characterized by peripheral vasospasm and ischemia of the extremities and other tissues. Hepatitis C virus (HCV):
Direct-Acting Antivirals: elbasvir/grazoprevir↑ elbasvir/grazoprevir Co-administration is contraindicated due to potential for the increased risk of alanine transaminase (ALT) elevations. glecaprevir/pibrentasvir ↑ glecaprevir
↑ pibrentasvirCo-administration of PREZCOBIX with glecaprevir/pibrentasvir is not recommended. Herbal product:
St. John's wort ( Hypericum perforatum)↓ darunavir
↓ cobicistatCo-administration is contraindicated due to potential for reduced plasma concentrations of darunavir, which may result in loss of therapeutic effect and development of resistance. Hormonal contraceptives: Additional or alternative (non-hormonal) forms of contraception should be considered when estrogen-containing contraceptives are co-administered with PREZCOBIX [see Use in Specific Populations (8.3)] . drospirenone/ethinylestradiol ↑ drospirenone
↓ ethinylestradiolFor co-administration with drospirenone, clinical monitoring is recommended due to the potential for hyperkalemia. Other progestin/estrogen contraceptives progestin: effects unknown
estrogen: effects unknownNo data are available to make recommendations on co-administration with other hormonal contraceptives. Immunosuppressants:
cyclosporine, sirolimus, tacrolimus↑ immunosuppressants These immunosuppressant agents are metabolized by CYP3A. Therapeutic drug monitoring is recommended with concomitant use Immunosuppressant /neoplastic:
everolimus↑ immunosuppressants
Co-administration of everolimus and PREZCOBIX is not recommended.irinotecan Discontinue PREZCOBIX at least 1 week prior to starting irinotecan therapy. Do not administer PREZCOBIX with irinotecan unless there are no therapeutic alternatives. Inhaled beta agonist:
salmeterol↑ salmeterol Co-administration with salmeterol is not recommended and may result in increased risk of cardiovascular adverse events associated with salmeterol, including QT prolongation, palpitations, and sinus tachycardia. Lipid Modifying Agents HMG-CoA reductase inhibitors:
lovastatin, simvastatin↑ lovastatin
↑ simvastatinCo-administration is contraindicated due to potential for serious reactions such as myopathy including rhabdomyolysis. atorvastatin, fluvastatin, pitavastatin, pravastatin, rosuvastatin ↑ atorvastatin
↑ fluvastatin
↑ pravastatin
↑ rosuvastatin
pitavastatin: effect unknownFor atorvastatin, fluvastatin, pitavastatin, pravastatin, and rosuvastatin, start with the lowest recommended dose and titrate while monitoring for safety (e.g. myopathy).
Dosage recommendations with atorvastatin or rosuvastatin are as follows:- atorvastatin dosage should not exceed 20 mg/day
- rosuvastatin dosage should not exceed 20 mg/day
Other lipid modifying agents:
lomitapide↑ lomitapide Co-administration is contraindicated due to potential for markedly increased transaminases associated with increased plasma concentrations of lomitapide. Narcotic analgesics metabolized by CYP3A:
e.g. fentanyl, oxycodone↑ fentanyl
↑ oxycodoneCareful monitoring of therapeutic effects and adverse reactions associated with CYP3A-metabolized narcotic analgesics (including potentially fatal respiratory depression) is recommended with co-administration. tramadol ↑ tramadol A dose decrease may be needed for tramadol with concomitant use. Narcotic analgesic for treatment of opioid dependence:
buprenorphine, buprenorphine/naloxone, methadonebuprenorphine or buprenorphine/ naloxone: effects unknown
methadone: effects unknownInitiation of buprenorphine, buprenorphine/naloxone or methadone in patients taking PREZCOBIX: Carefully titrate the dose of buprenorphine, buprenorphine/naloxone or methadone to the desired effect; use the lowest feasible initial or maintenance dose.
Initiation of PREZCOBIX in patients taking buprenorphine, buprenorphine/naloxone or methadone: A dose adjustment for buprenorphine, buprenorphine/naloxone or methadone may be needed. Monitor clinical signs and symptoms.Opioid Antagonist naloxegol ↑ naloxegol Co-administration of PREZCOBIX and naloxegol is contraindicated due to potential for precipitating opioid withdrawal symptoms. Phosphodiesterase PDE-5 inhibitors:
e.g. avanafil, sildenafil, tadalafil, vardenafil↑ PDE-5 inhibitors Co-administration with avanafil is not recommended because a safe and effective avanafil dosage regimen has not been established.
Co-administration with PDE-5 inhibitors may result in an increase in PDE-5 inhibitor-associated adverse reactions including hypotension, syncope, visual disturbances and priapism.
Use of PDE-5 inhibitors for pulmonary arterial hypertension (PAH):
Co-administration with sildenafil used for PAH is contraindicated due to potential for sildenafil associated adverse reactions (which include visual disturbances, hypotension, prolonged erection, and syncope).
The following dose adjustments are recommended for use of tadalafil with PREZCOBIX:- Initiation of tadalafil in patients taking PREZCOBIX: In patients receiving PREZCOBIX for at least one week, start tadalafil at 20 mg once daily. Increase to 40 mg once daily based upon individual tolerability.
- Initiation of PREZCOBIX in patients taking tadalafil: Avoid use of tadalafil during the initiation of PREZCOBIX. Stop tadalafil at least 24 hours prior to starting PREZCOBIX. After at least one week following the initiation of PREZCOBIX, resume tadalafil at 20 mg once daily. Increase to 40 mg once daily based upon individual tolerability.
- Patients switching from darunavir co-administered with ritonavir to PREZCOBIX: Maintain tadalafil dose.
Sildenafil at a single dose not exceeding 25 mg in 48 hours, vardenafil at a single dose not exceeding 2.5 mg dose in 72 hours, or tadalafil at a single dose not exceeding 10 mg dose in 72 hours can be used with increased monitoring for PDE-5 inhibitor-associated adverse reactions.Platelet aggregation inhibitor: ticagrelor ↑ ticagrelor Co-administration of PREZCOBIX and ticagrelor is not recommended. clopidogrel ↓ clopidogrel active metabolite Co-administration of PREZCOBIX with clopidogrel is not recommended due to the potential reduction of the antiplatelet activity of clopidogrel. prasugrel ↔ prasugrel active metabolite No dose adjustment is needed when prasugrel is co-administered with PREZCOBIX. Sedatives/hypnotics:
orally administered midazolam, triazolam↑ midazolam
↑ triazolamCo-administration is contraindicated due to potential for serious and/or life-threatening reactions such as prolonged or increased sedation or respiratory depression. Triazolam and orally administered midazolam are extensively metabolized by CYP3A. Co-administration of triazolam or orally administered midazolam with PREZCOBIX may cause large increases in the concentrations of these benzodiazepines. metabolized by CYP3A:
e.g. buspirone, diazepam, estazolam, zolpidem↑ sedatives/hypnotics With concomitant use, titration is recommended with sedatives/hypnotics metabolized by CYP3A and a lower dose of the sedatives/hypnotics should be considered with monitoring for increased and prolonged effects or adverse reactions. parenterally administered midazolam Co-administration of parenteral midazolam should be done in a setting that ensures close clinical monitoring and appropriate medical management in case of respiratory depression and/or prolonged sedation. Dose reduction for parenteral midazolam should be considered, especially if more than a single dose of midazolam is administered. Urinary antispasmodics fesoterodine ↑ fesoterodine When fesoterodine is co-administered with PREZCOBIX, do not exceed a fesoterodine dose of 4 mg once daily. solifenacin ↑ solifenacin When solifenacin is co-administered with PREZCOBIX, do not exceed a solifenacin dose of 5 mg once daily. 7.4 Drugs without Clinically Significant Interactions with PREZCOBIX
Clinically relevant drug-drug interactions have not been observed or are not anticipated with concomitant use of darunavir and cobicistat with rilpivirine, dolutegravir, raltegravir, abacavir, emtricitabine, emtricitabine/tenofovir alafenamide, tenofovir DF, lamivudine, stavudine, zidovudine, or acid modifying medications (antacids, H 2-receptor antagonists, proton pump inhibitors).
-
8 USE IN SPECIFIC POPULATIONS
8.1 Pregnancy
Pregnancy Exposure Registry
There is a pregnancy exposure registry that monitors pregnancy outcomes in individuals exposed to PREZCOBIX during pregnancy. Healthcare providers are encouraged to register patients by calling the Antiretroviral Pregnancy Registry (APR) 1-800-258-4263.
Risk Summary
PREZCOBIX is not recommended during pregnancy because of substantially lower exposures of darunavir and cobicistat during the second and third trimesters [see Dosage and Administration (2.5)] . A study evaluating the pharmacokinetics of antiretrovirals during pregnancy demonstrated substantially lower exposures of darunavir and cobicistat in the second and third trimesters compared to the post-partum period (see Data) and [see Clinical Pharmacology (12.3)].
Prospective pregnancy data from the APR are not sufficient to adequately assess the risk of birth defects or miscarriage. However, available data from the APR show no statistically significant difference in the overall risk of major birth defects for darunavir and cobicistat compared with the background rate for major birth defects of 2.7% in a U.S. reference population of the Metropolitan Atlanta Congenital Defects Program (MACDP) (see Data) . The rate of miscarriage is not reported in the APR. The estimated background rate of miscarriage in clinically recognized pregnancies in the U.S. general population is 15–20%. The background risk of major birth defects and miscarriage for the indicated population is unknown.
In animal reproduction studies, no adverse developmental effects were observed when the components of PREZCOBIX were administered separately at darunavir exposures less than 1 (mice and rabbits) and 3-times (rats), and at cobicistat exposures 1.6 (rats) and 3.8 (rabbits) times human exposures at the recommended daily dose of these components in PREZCOBIX (see Data) . No adverse developmental effects were seen when cobicistat was administered to rats through lactation at cobicistat exposures up to 1.2 times the human exposure at the recommended therapeutic dose.
Clinical Considerations
Not Recommended During Pregnancy
PREZCOBIX is not recommended for use during pregnancy because of substantially lower exposures of darunavir and cobicistat during pregnancy (see Data) and [see Clinical Pharmacology (12.3)] .
PREZCOBIX should not be initiated in pregnant individuals. An alternative regimen is recommended for individuals who become pregnant during therapy with PREZCOBIX.
Data
Human Data
PREZCOBIX in combination with a background regimen was evaluated in a clinical trial of 7 pregnant participants taking PREZCOBIX prior to enrollment and who were willing to remain on PREZCOBIX throughout the study. The study period included the second and third trimesters, and through 12 weeks postpartum. Six pregnant participants completed the trial.
Exposure to darunavir and cobicistat as part of an antiretroviral regimen was substantially lower during the second and third trimesters of pregnancy compared with postpartum [see Clinical Pharmacology (12.3)] .
One out of 6 pregnant participants who completed the study experienced virologic failure with HIV-1 RNA >1,000 copies/mL from the third trimester visit through the postpartum period. Five pregnant participants had sustained virologic response (HIV RNA <50 copies/mL) throughout the study period. There are no clinical data on the virologic response when PREZCOBIX is initiated during pregnancy.
Prospective reports from the APR of overall major birth defects in pregnancies exposed to the components of PREZCOBIX are compared with a U.S. background major birth defect rate. Methodological limitations of the APR include the use of MACDP as the external comparator group. Limitations of using an external comparator include differences in methodology and populations, as well as confounding due to the underlying disease.
There were no new clinically relevant safety findings compared with the known safety profile of PREZCOBIX in adults with HIV-1.
Darunavir: Based on prospective reports to the APR of over 980 exposures to darunavir-containing regimens during pregnancy resulting in live births (including over 660 exposed in the first trimester and over 320 exposed in the second/third trimester), the prevalence of birth defects in live births was 3.6% (95% CI: 2.3% to 5.3%) with first trimester exposure to darunavir-containing regimens and 2.5% (95% CI: 1.1% to 4.8%) with second/third trimester exposure to darunavir-containing regimens.
Cobicistat: Based on prospective reports to the APR of over 570 exposures to cobicistat-containing regimens during pregnancy resulting in live births (including over 480 exposed in the first trimester and over 80 exposed in the second/third trimester), the prevalence of birth defects in live births was 3.7% (95% CI: 2.2% to 5.7%) and 1.1% (95% CI: 0.0% to 6.2%) with first and second/third trimester, respectively, to cobicistat-containing regimens.
Animal Data
Darunavir: Reproduction studies conducted with darunavir showed no embryotoxicity or teratogenicity in mice (doses up to 1000 mg/kg from gestation day (GD) 6–15 with darunavir alone) and rats (doses up to 1000 mg/kg from GD 7–19 in the presence or absence of ritonavir) as well as in rabbits (doses up to 1000 mg/kg/day from GD 8–20 with darunavir alone). In these studies, darunavir exposures (based on AUC) were higher in rats (3-fold), whereas in mice and rabbits, exposures were lower (less than 1-fold) compared to those obtained in humans at the recommended clinical dose of darunavir co-administered with ritonavir.
Cobicistat: Cobicistat was administered orally to pregnant rats at doses up to 125 mg/kg/day on GD 6–17. Increases in post-implantation loss and decreased fetal weights were observed at a maternal toxic dose of 125 mg/kg/day. No malformations were noted at doses up to 125 mg/kg/day. Systemic exposures (AUC) at 50 mg/kg/day in pregnant females were 1.6 times higher than human exposures at the recommended daily dose of cobicistat.
In pregnant rabbits, cobicistat was administered orally at doses up to 100 mg/kg/day during GD 7–20. No maternal or embryo/fetal effects were noted at the highest dose of 100 mg/kg/day. Systemic exposures (AUC) at 100 mg/kg/day were 3.8 times higher than human exposures at the recommended daily dose of cobicistat.
In a pre/postnatal developmental study in rats, cobicistat was administered orally at doses up to 75 mg/kg from GD 6 to postnatal day 20, 21, or 22. At doses of 75 mg/kg/day, neither maternal nor developmental toxicity was noted. Systemic exposures (AUC) at this dose were 1.2 times the human exposures at the recommended daily dose of cobicistat.
8.2 Lactation
Risk Summary
There are no data on the presence of darunavir or cobicistat in human milk, the effects on the breastfed infant, or the effects on milk production. Darunavir and cobicistat are present in the milk of lactating rats (see Data) . Potential risks of breastfeeding include: (1) HIV-1 transmission (in infants without HIV-1), (2) developing viral resistance (in infants with HIV-1), and (3) serious adverse reactions in breastfed infant similar to those seen in adults.
Data
Animal Data
Darunavir: Studies in rats (with darunavir alone or with ritonavir) have demonstrated that darunavir is excreted in milk. In the rat pre- and postnatal development study, a reduction in pup body weight gain was observed due to exposure of pups to drug substances via milk. The maximal maternal plasma exposures achieved with darunavir (up to 1000 mg/kg with ritonavir) were approximately 50% of those obtained in humans at the recommended clinical dose of darunavir with ritonavir.
8.3 Females and Males of Reproductive Potential
Contraception
Additional or alternative (non-hormonal) forms of contraception should be considered when estrogen-containing contraceptives are co-administered with PREZCOBIX. For co-administration with drospirenone, clinical monitoring is recommended due to the potential for hyperkalemia. No data are available to make recommendations on co-administration with other hormonal contraceptives [see Drug Interactions (7.3)].
8.4 Pediatric Use
The safety and effectiveness of PREZCOBIX for the treatment of HIV-1 in pediatric patients weighing at least 25 kg was established through a trial with components of PREZCOBIX. Use of PREZCOBIX in this group is supported by evidence from adequate and well-controlled studies in adults with additional pharmacokinetic, safety, and virologic data from a study of components of PREZCOBIX (Trial GS-US-216-0128) in pediatric participants with HIV-1 aged 6 to less than 18 years [see Adverse Reactions (6.1), Clinical Pharmacology (12.3), and Clinical Studies (14.2)] .
The safety and effectiveness of PREZCOBIX have not been established in pediatric patients weighing less than 25 kg. Darunavir, a component of PREZCOBIX is not recommended in pediatric patients below 3 years of age because of toxicity and mortality observed in juvenile rats dosed with darunavir.
Juvenile Animal Toxicity Data
Darunavir: In a juvenile toxicity study where rats were directly dosed with darunavir (up to 1000 mg/kg), deaths occurred from post-natal day 5 at plasma exposure levels ranging from 0.1 to 1.0 of the human exposure levels. In a 4-week rat toxicology study, when dosing was initiated on post-natal day 23 (the human equivalent of 2 to 3 years of age), no deaths were observed with a plasma exposure (in combination with ritonavir) 2 times the human plasma exposure levels.
8.5 Geriatric Use
Clinical trials of PREZCOBIX did not include sufficient numbers of patients aged 65 and over to determine whether they respond differently from younger patients. In general, caution should be exercised in the administration and monitoring of PREZCOBIX in elderly patients, reflecting the greater frequency of decreased hepatic function, and of concomitant disease or other drug therapy [see Clinical Pharmacology (12.3)] .
8.6 Hepatic Impairment
No clinical trials were conducted with darunavir co-administered with cobicistat in hepatically impaired patients and the effect of hepatic impairment on darunavir exposure when co-administered with cobicistat has not been evaluated. Based on the recommendations for darunavir co-administered with ritonavir, a dose adjustment for patients with mild or moderate hepatic impairment is not necessary. No pharmacokinetic or safety data are available regarding the use of darunavir in patients with severe hepatic impairment. Therefore, PREZCOBIX is not recommended for use in patients with severe hepatic impairment [see Clinical Pharmacology (12.3)] .
8.7 Renal Impairment
A renal impairment trial was not conducted for darunavir co-administered with cobicistat [see Clinical Pharmacology (12.3)] . Cobicistat has been shown to decrease estimated creatinine clearance without affecting actual renal glomerular function. Dosing recommendations are not available for drugs that require dosage adjustment for renal impairment when used in combination with PREZCOBIX [see Warnings and Precautions (5.3)and Clinical Pharmacology (12.2)] .
-
10 OVERDOSAGE
Human experience of acute overdose with PREZCOBIX is limited. No specific antidote is available for overdose with PREZCOBIX. Treatment of overdose with PREZCOBIX consists of general supportive measures including monitoring of vital signs and observation of the clinical status of the patient. Since both darunavir and cobicistat are highly protein bound, dialysis is unlikely to be beneficial in significant removal of the active substance.
-
11 DESCRIPTION
PREZCOBIX ® is a fixed-dose combination tablet containing darunavir and cobicistat. Darunavir is an inhibitor of the human immunodeficiency virus (HIV-1) protease. Cobicistat is a mechanism-based inhibitor of cytochrome P450 (CYP) enzymes of the CYP3A family.
PREZCOBIX ® 800 mg darunavir/150 mg cobicistat tablets are for oral administration. Each tablet contains darunavir ethanolate equivalent to 800 mg of darunavir and 150 mg of cobicistat. The tablets include the following inactive ingredients: colloidal silicon dioxide, crospovidone, hypromellose, magnesium stearate, and silicified microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide red, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide.
PREZCOBIX ® 675 mg darunavir/150 mg cobicistat tablets are for oral administration. Each tablet contains darunavir ethanolate equivalent to 675 mg of darunavir and 150 mg of cobicistat. The tablets include the following inactive ingredients: colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide yellow, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide.
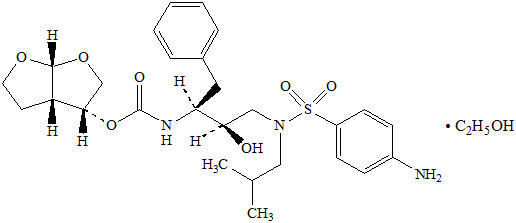
Darunavir: Darunavir, in the form of darunavir ethanolate, has the following chemical name: [(1 S,2 R)-3-[[(4-aminophenyl)sulfonyl](2-methylpropyl)amino]-2-hydroxy-1-(phenylmethyl)propyl]-carbamic acid (3 R,3a S,6a R)-hexahydrofuro[2,3- b]furan-3-yl ester monoethanolate. Its molecular formula is C 27H 37N 3O 7S ∙ C 2H 5OH and its molecular weight is 593.73. Darunavir ethanolate has the following structural formula:
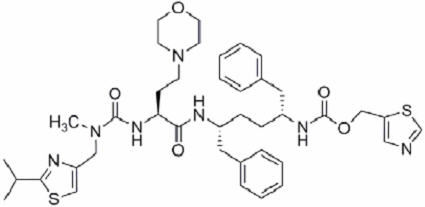
Cobicistat: Cobicistat is adsorbed onto silicon dioxide. The chemical name for cobicistat is 1,3-thiazol-5-ylmethyl[(2 R, 5 R)-5-{[(2 S)2-[(methyl{[2-(propan-2-yl)-1,3-thiazol-4-yl]methyl}carbamoyl)amino]-4-(morpholin-4yl)butanoyl]amino}-1,6-diphenylhexan-2-yl] carbamate. It has a molecular formula of C 40 H 53 N 7 O 5 S 2 and a molecular weight of 776.0. It has the following structural formula:
-
12 CLINICAL PHARMACOLOGY
12.1 Mechanism of Action
PREZCOBIX is a fixed-dose combination of an HIV-1 antiviral drug, darunavir and a CYP3A inhibitor, cobicistat [see Microbiology (12.4)].
12.2 Pharmacodynamics
Cardiac Electrophysiology
Separate thorough QT trials have been conducted for darunavir co-administered with ritonavir and for cobicistat. The effect of darunavir co-administered with cobicistat on the QT interval has not been evaluated.
Darunavir: In a thorough QT/QTc study in 40 healthy participants, darunavir doses (co-administered with 100 mg ritonavir) of approximately 2 times the recommended darunavir dose did not affect the QT/QTc interval.
Cobicistat: The effect of a single dose of cobicistat 250 mg and 400 mg (approximately 1.7 and 2.7 times the recommended dose) on QTc interval was evaluated in a randomized, placebo- and active-controlled (moxifloxacin 400 mg) four-period crossover thorough QT trial in 48 healthy participants. In this trial, no significant QTc prolongation effect of cobicistat was detected. The dose of 400 mg cobicistat is expected to provide information on a high exposure clinical scenario. Prolongation of the PR interval was noted in participants receiving cobicistat in the same trial. The maximum mean (95% upper confidence bound) difference in PR from placebo after baseline-correction was 9.5 (12.1) msec for 250 mg and 20.2 (22.8) msec for 400 mg of cobicistat .
Effects on Serum Creatinine
Cobicistat: The effect of cobicistat on serum creatinine was investigated in a trial in participants with normal renal function (eGFR ≥80 mL/min, N=12) and mild-to-moderate renal impairment (eGFR 50–79 mL/min, N=18). A statistically significant decrease in the estimated glomerular filtration rate, calculated by Cockcroft-Gault method (eGFR CG) from baseline, was observed after 7 days of treatment with cobicistat 150 mg among participants with normal renal function (-9.9 ± 13.1 mL/min) and mild-to-moderate renal impairment (-11.9 ± 7.0 mL/min). No statistically significant changes in eGFR CGwere observed compared to baseline for participants with normal renal function or mild-to-moderate renal impairment 7 days after cobicistat was discontinued. The actual glomerular filtration rate, as determined by the clearance of probe drug iohexol, was not altered from baseline following treatment of cobicistat among participants with normal renal function and mild-to-moderate renal impairment, indicating that cobicistat inhibits tubular secretion of creatinine, reflected as a reduction in eGFR CG, without affecting the actual glomerular filtration rate.
12.3 Pharmacokinetics
The pharmacokinetics of darunavir co-administered with cobicistat (150 mg) have been evaluated in healthy adults and in adults with HIV-1.
Darunavir is primarily metabolized by CYP3A. Cobicistat inhibits CYP3A, thereby increasing the plasma concentrations of darunavir.
Under fed (535 total kcal, 171 kcal from fat, 268 kcal from carbohydrates, 96 kcal from protein) and fasted conditions in healthy participants, the 90% confidence intervals when comparing darunavir exposure between PREZCOBIX (800 mg/150 mg) and darunavir 800 mg co-administered with cobicistat 150 mg as single entities were within 80–125%. No clinically significant difference was observed between PREZCOBIX (675 mg/150 mg) and darunavir 675 mg co-administered with cobicistat 150 mg as single entities under fed conditions.
Darunavir exposure when comparing darunavir co-administered with cobicistat (as single entities) to darunavir co-administered with ritonavir was evaluated in a relative bioavailability trial [see cobicistat full prescribing information].Table 3 displays the pharmacokinetic estimates of darunavir after oral administration of darunavir 800 mg co-administered with ritonavir 100 mg once daily (based on sparse sampling in 335 participants in Trial TMC114-C211 and 280 participants in Trial TMC114-C229) and darunavir 800 mg co-administered with cobicistat 150 mg once daily administered as single entities (based on sparse sampling in 298 participants in Trial GS-US-216-0130) to HIV-1 participants.
Table 3: Pharmacokinetic Estimates of Darunavir as Darunavir 800 mg Co-administered with Ritonavir 100 mg Once Daily (Trial TMC114-C211, 48 Week Analysis and Trial TMC114-C229, 48 Week Analysis) and Darunavir 800 mg Co-administered with Cobicistat 150 mg Once Daily (Trial GS-US-216-0130, 24 Week Analysis) Trial TMC114-C211
(treatment-naïve)
Darunavir 800 mg co-administered with ritonavir 100 mg once dailyTrial TMC114-C229
(treatment-experienced)
Darunavir 800 mg co-administered with ritonavir 100 mg once dailyTrial GS-US-216-0130
(treatment-naïve and experienced)
Darunavir 800 mg co-administered with cobicistat 150 mg once dailyParameter N=335 N=280 N=298 AUC 0–24h=area under the concentration-time curve over a 24-hour dosing interval at steady state; C 0h=plasma concentration at the end of a 24-hour dosing interval at steady state; N=number of participants; SD=standard deviation AUC 0–24h(ng∙h/mL) Mean ± SD 93026 ± 27050 93334 ± 28626 100152 ± 32042 Median (Range) 87854 (45000–219240) 87788 (45456–236920) 96900 (34500–224000) C 0h(ng/mL) Mean ± SD 2282 ± 1168 2160 ± 1201 2043 ± 1257 Median (Range) 2041 (368–7242) 1896 (184–7881) 1875 (70–6890) Absorption and Bioavailability
In healthy participants, under fed conditions, when single doses of the darunavir and cobicistat fixed-dose combination tablet were administered, the maximum plasma concentration was achieved within approximately 4 to 4.5 hours for darunavir and approximately 4 to 5 hours for cobicistat.
Effects of Food on Oral Absorption
When compared to fasted conditions, administration of PREZCOBIX to healthy adult participants with a high-fat meal (965 total kcal: 129 kcal from protein, 236 kcal from carbohydrates and 600 kcal from fat) resulted in a 70% increase in AUC (0–inf)and a 127% increase in C maxfor darunavir. Cobicistat exposures were not affected by food. PREZCOBIX should be taken with food.
Distribution
Metabolism
Darunavir: In vitro experiments with human liver microsomes (HLMs) indicate that darunavir primarily undergoes oxidative metabolism. Darunavir is extensively metabolized by CYP enzymes, primarily by CYP3A. A mass balance trial in healthy participants showed that after single dose administration of 400 mg 14C-darunavir co-administered with 100 mg ritonavir, the majority of the radioactivity in the plasma was due to darunavir. At least 3 oxidative metabolites of darunavir have been identified in humans; all showed activity that was at least 90% less than the activity of darunavir against wild-type HIV-1.
Elimination
Darunavir: A mass balance trial in healthy participants showed that after single dose administration of 400 mg 14C-darunavir co-administered with 100 mg ritonavir, approximately 79.5% and 13.9% of the administered dose of 14C-darunavir was recovered in the feces and urine, respectively. Unchanged darunavir accounted for approximately 41.2% and 7.7% of the administered dose in feces and urine, respectively.
When single doses of the darunavir and cobicistat fixed-dose combination tablet were administered, the terminal elimination half-life of darunavir was approximately 7 hours under fed conditions.
Cobicistat: When single doses of the darunavir and cobicistat fixed-dose combination tablet were administered, the terminal elimination half-life of cobicistat was approximately 4 hours under fed conditions. With single dose administration of 14C-cobicistat after multiple dosing of cobicistat for six days, the mean percent of the administered dose excreted in feces and urine was 86.2% and 8.2%, respectively.
Specific Populations
Hepatic Impairment
Darunavir: Darunavir is primarily metabolized by the liver. The steady-state pharmacokinetic parameters of darunavir were similar after multiple dose co-administration of darunavir 600 mg co-administered with ritonavir 100 mg twice daily to participants with normal hepatic function (n=16), mild hepatic impairment (Child-Pugh Class A, n=8), and moderate hepatic impairment (Child-Pugh Class B, n=8). The effect of severe hepatic impairment on the pharmacokinetics of darunavir has not been evaluated [see Use in Specific Populations (8.6)].
Cobicistat: Cobicistat is primarily metabolized by the liver. A trial evaluating the pharmacokinetics of cobicistat was performed in participants with moderate hepatic impairment. No clinically relevant differences in cobicistat pharmacokinetics were observed between participants with moderate hepatic impairment (Child-Pugh Class B) and healthy participants. The effect of severe hepatic impairment on the pharmacokinetics of cobicistat has not been evaluated [see Use in Specific Populations (8.6)].
Hepatitis B or Hepatitis C Virus Co-Infection
Darunavir: In participants with HIV-1 taking darunavir co-administered with ritonavir, the 48 week analysis of the data from clinical studies in participants with HIV-1 indicated that hepatitis B and/or hepatitis C virus co-infection status had no apparent effect on the exposure of darunavir.
The effect of hepatitis B and/or C virus infection on the pharmacokinetics of PREZCOBIX have not been evaluated.
Renal Impairment
Darunavir: Population pharmacokinetic analysis showed that the pharmacokinetics of darunavir were not significantly affected in participants with HIV-1 with moderate renal impairment taking darunavir co-administered with ritonavir (creatinine clearance between 30–60 mL/min, n=20). There are no pharmacokinetic data available in participants with HIV-1 with severe renal impairment or end stage renal disease taking darunavir co-adminstered with either ritonavir or cobicistat [see Use in Specific Populations (8.7)].
Cobicistat: A trial of the pharmacokinetics of cobicistat was performed in non-HIV participants with severe renal impairment (estimated creatinine clearance below 30 mL/min). No clinically relevant differences in cobicistat pharmacokinetics were observed between participants with severe renal impairment and healthy participants [see Use in Special Populations (8.7)].
Gender
Race
Geriatric Patients
Darunavir: In participants with HIV-1 taking darunavir co-administered with ritonavir, population pharmacokinetic analysis showed no considerable differences in darunavir pharmacokinetics for ages 18 to 75 years compared to ages greater than or equal to 65 years (n=12) [see Use in Specific Populations (8.5)] .
Pediatrics Weighing at Least 25 kg
Available pharmacokinetic data for the different components of PREZCOBIX indicate that there were no clinically relevant differences in darunavir exposure between adults and adolescents or children weighing at least 25 kg (see Table 4). Cobicistat exposures were similar between adults and adolescents but higher in children aged 6 to less than 12 years, weighing at least 25 kg. The difference observed in children for cobicistat is not considered clinically relevant.
Table 4: Pharmacokinetic Estimates Comparison of Darunavir AUC 0–24hand C 0hin Pediatric and Adult Studies Parameter GS-US-216-0128 in Participants Aged ≥ 6 to < 12 Years and Weighing ≥ 25 to < 40 kg
DRV 675 mg/COBI 150 mg
N=8GS-US-216-0128 in Participants Aged ≥ 12 to < 18 Years and Weighing ≥ 40 kg
DRV 800 mg/COBI 150 mg
N=8TMC114FD2HTX3001 in Adult Participants
DRV 800 mg/COBI 150 mg/FTC 200 mg/TAF 10 mg
N=356AUC 0–24h=area under the concentration-time curve over a 24-hour dosing interval at steady state; C 0h=plasma concentration at the end of a 24-hour dosing interval at steady state; COBI=cobicistat; DRV=darunavir; FTC=emtricitabine; N=number of participants; TAF=tenofovir alafenamide - *
- Mean ± standard deviation
AUC 0–24h(ng ∙ h/mL)* 92,052 ±17,254 69,474 ±10,359 87,909 ±20,232 C 0h(ng/mL) * 1,345 ±569 938 ±445 1,899 ±759 Pregnancy and Postpartum
The exposure to total and unbound darunavir boosted with cobicistat after intake of PREZCOBIX as part of an antiretroviral regimen was substantially lower during the second and third trimesters of pregnancy compared with 6–12 weeks postpartum (see Table 5 and Figure 1).
Table 5: Pharmacokinetic Results of Total Darunavir after Administration of PREZCOBIX Once Daily as Part of an Antiretroviral Regimen, During the 2 nd Trimester of Pregnancy, the 3 rd Trimester of Pregnancy, and Postpartum Parameter 2 ndTrimester of pregnancy
N=73 rdTrimester of pregnancy
N=6Postpartum
(6–12 weeks)
N=6C max=maximum plasma concentration at steady state; AUC 0–24h=area under the concentration-time curve over a 24-hour dosing interval at steady state; C min=minimum plasma concentration at steady state - *
- Mean ± standard deviation
C max(ng/mL) * 4340 ± 1616 4910 ± 970 7918 ± 2199 AUC 0–24h(ng∙h/mL) * 47293 ± 19058 47991 ± 9879 99613 ± 34862 C min(ng/mL) * 168 ± 149 184 ± 99 1538 ± 1344 Figure 1: Pharmacokinetic Results (Within-Subject Comparison) of Total and Unbound Darunavir and Total Cobicistat after Administration of PREZCOBIX at 800/150 mg Once Daily as Part of an Antiretroviral Regimen, During the 2 ndand 3 rdTrimester of Pregnancy Compared to Postpartum
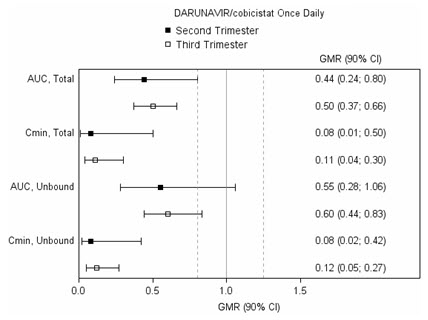
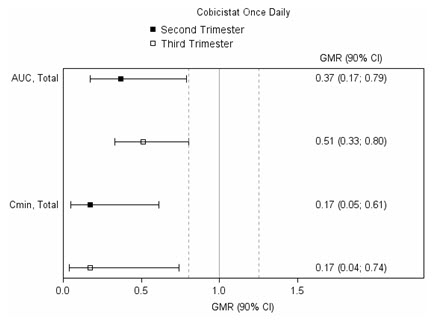
Legend: 90% CI: 90% confidence interval; GMR: geometric mean ratio (i.e. second or third trimester / postpartum). Solid vertical line: ratio of 1.0; dotted vertical lines: reference lines of 0.8 and 1.25.
Drug Interactions
Darunavir is metabolized by CYP3A. Cobicistat is metabolized by CYP3A and, to a minor extent, by CYP2D6. Darunavir co-administered with cobicistat is an inhibitor of CYP3A and CYP2D6. Cobicistat inhibits the following transporters: P-gp, BCRP, MATE1, OATP1B1, and OATP1B3. Based on in vitro data, cobicistat is not expected to induce CYP1A2 or CYP2B6 and based on in vivo data, cobicistat is not expected to induce MDR1 or, in general, CYP3A to a clinically significant extent. The induction effect of cobicistat on CYP2C9, CYP2C19, or UGT1A1 is unknown, but is expected to be low based on CYP3A in vitro induction data [see Drug Interactions (7)] .
A drug-drug interaction study between darunavir/cobicistat and dabigatran etexilate was conducted in healthy participants. The effects of darunavir on co-administration with dabigatran etexilate are summarized in Table 6.
Table 6: Drug Interactions: Pharmacokinetic Parameters for Co-Administered Drugs in the Presence of darunavir/cobicistat Co-administered drug Dose/Schedule N PK LS Mean ratio (90% CI) of
co-administered drug
pharmacokinetic parameters with/without darunavir
no effect =1.00Co-administered drug Darunavir/ cobicistat C max AUC C min N = number of participants with data
q.d. = once daily- *
- 800/150 mg q.d. for 14 days before co-administered with dabigatran etexilate.
Dabigatran etexilate 150 mg 800/150 mg single dose 14 ↑ 2.64
(2.29–3.05)2.64
(2.32–3.00)- 800/150 mg q.d. * 14 ↑ 1.99
(1.72–2.30)1.88
(1.65–2.13)- 12.4 Microbiology
Mechanism of Action
Antiviral Activity
Darunavir: Darunavir exhibits activity against laboratory strains and clinical isolates of HIV-1 and laboratory strains of HIV-2 in acutely infected T-cell lines, human peripheral blood mononuclear cells, and human monocytes/macrophages with median EC 50values ranging from 1.2 to 8.5 nM (0.7 to 5.0 ng/mL). Darunavir demonstrates antiviral activity in cell culture against a broad panel of HIV-1 group M (A, B, C, D, E, F, G), and group O primary isolates with EC 50values ranging from less than 0.1 to 4.3 nM. The EC 50value of darunavir increases by a median factor of 5.4 in the presence of human serum. Darunavir did not show antagonism when studied in combination with the HIV protease inhibitors (PIs) amprenavir, atazanavir, indinavir, lopinavir, nelfinavir, ritonavir, saquinavir, or tipranavir, the N(t)RTIs abacavir, didanosine, emtricitabine, lamivudine, stavudine, tenofovir, zalcitabine, or zidovudine, the NNRTIs delavirdine, efavirenz, etravirine, rilpivirine, or nevirapine, and the fusion inhibitor enfuvirtide.
Resistance
Cell Culture
Darunavir: HIV-1 isolates with a decreased susceptibility to darunavir have been selected in cell culture and obtained from participants treated with darunavir co-administered with ritonavir. Darunavir-resistant virus derived in cell culture from wild-type HIV-1 had 21- to 88-fold decreased susceptibility to darunavir and developed 2 to 4 of the following amino acid substitutions S37D, R41E/T, K55Q, H69Q, K70E, T74S, V77I, or I85V in the protease. Selection in cell culture of darunavir resistant HIV-1 from nine HIV-1 strains harboring multiple PI resistance-associated substitutions resulted in the overall emergence of 22 mutations in the protease gene, coding for amino acid substitutions L10F, V11I, I13V, I15V, G16E, L23I, V32I, L33F, S37N, M46I, I47V, I50V, F53L, L63P, A71V, G73S, L76V, V82I, I84V, T91A/S, and Q92R, of which L10F, V32I, L33F, S37N, M46I, I47V, I50V, L63P, A71V, and I84V were the most prevalent. These darunavir-resistant viruses had at least eight protease substitutions and exhibited 50- to 641-fold decreases in darunavir susceptibility with final EC 50values ranging from 125 nM to 3461 nM.
Clinical Studies
The resistance profile of PREZCOBIX is driven by darunavir. Cobicistat does not select any HIV resistance substitutions, due to its lack of antiviral activity. For the clinical resistance profile of darunavir, refer to the darunavir full prescribing information.
Cross-resistance
Cross-resistance among PIs has been observed. Darunavir has a less than 10-fold decreased susceptibility in cell culture against 90% of 3309 clinical isolates resistant to amprenavir, atazanavir, indinavir, lopinavir, nelfinavir, ritonavir, saquinavir, and/or tipranavir showing that viruses resistant to these PIs remain susceptible to darunavir. A less than 10-fold decreased susceptibility was observed for the other PIs in 26% to 96% of these PI resistant clinical isolates [nelfinavir (26%), ritonavir (34%), lopinavir (46%), indinavir (57%), atazanavir (59%), saquinavir (64%), amprenavir (70%), and tipranavir (96%)].
Cross-resistance between darunavir and nucleoside/nucleotide reverse transcriptase inhibitors, non-nucleoside reverse transcriptase inhibitors, fusion inhibitors, CCR5 co-receptor antagonists, or integrase strand transfer inhibitors is unlikely because the viral targets are different.
Baseline Genotype/Phenotype and Virologic Outcome Analyses
Baseline International AIDS Society (IAS)-defined PI resistance substitutions confer reduced virologic response to darunavir. Please refer to the "Baseline Genotype/Phenotype and Virologic Outcome Analyses" section in the darunavir full prescribing information.
-
13 NONCLINICAL TOXICOLOGY
13.1 Carcinogenesis, Mutagenesis, Impairment of Fertility
Carcinogenesis and Mutagenesis
Darunavir: Darunavir was evaluated for carcinogenic potential by oral gavage administration to mice and rats up to 104 weeks. Daily doses of 150, 450 and 1000 mg/kg were administered to mice and doses of 50, 150 and 500 mg/kg were administered to rats. A dose-related increase in the incidence of hepatocellular adenomas and carcinomas was observed in males and females of both species and an increase in thyroid follicular cell adenomas was observed in male rats. The observed hepatocellular findings in rodents are considered to be of limited relevance to humans. Repeated administration of darunavir to rats caused hepatic microsomal enzyme induction and increased thyroid hormone elimination, which predispose rats but not humans, to thyroid neoplasms. At the highest tested doses, the systemic exposures to darunavir (based on AUC) were between 0.4- and 0.7-fold (mice) and 0.7- and 1-fold (rats) of exposures observed in humans at the recommended therapeutic doses (darunavir 600 mg co-administered with ritonavir 100 mg twice daily or darunavir 800 mg co-administered with ritonavir 100 mg once daily).
Darunavir was not mutagenic or genotoxic in a battery of in vitro and in vivo assays including bacterial reverse mutation (Ames), chromosomal aberration in human lymphocytes, and in vivo micronucleus test in mice.
Cobicistat: In a long-term carcinogenicity study in mice, no drug-related increases in tumor incidence were observed at doses up to 50 and 100 mg/kg/day in males and females, respectively. Cobicistat exposures at these doses were approximately 7 (male) and 16 (females) times, respectively, the human systemic exposure at the therapeutic daily dose. In a long-term carcinogenicity study of cobicistat in rats, an increased incidence of follicular cell adenomas and/or carcinomas in the thyroid gland was observed at doses of 25 and 50 mg/kg/day in males, and at 30 mg/kg/day in females. The follicular cell findings are considered to be rat-specific, secondary to hepatic microsomal enzyme induction and thyroid hormone imbalance, and are not relevant for humans. At the highest doses tested in the rat carcinogenicity study, systemic exposures were approximately 2 times the human systemic exposure at the therapeutic daily dose.
Cobicistat was not genotoxic in the reverse mutation bacterial test (Ames test), mouse lymphoma or rat micronucleus assays.
Impairment of Fertility
Darunavir: No effects on fertility or early embryonic development were observed with darunavir in rats.
Cobicistat: Cobicistat did not affect fertility in male or female rats at daily exposures (AUC) approximately 4-fold higher than human exposures at the recommended 150 mg daily dose.
Fertility was normal in the offspring of rats exposed daily from before birth ( in utero) through sexual maturity at daily exposures (AUC) of approximately 1.2-fold higher than human exposures at the recommended 150 mg daily dose.
-
14 CLINICAL STUDIES
14.1 Clinical Trial Results in Adults with HIV-1
The efficacy of PREZCOBIX in adults with HIV-1 is based on efficacy demonstrated in clinical trials of darunavir co-administered with ritonavir [see darunavir full prescribing information].
14.2 Clinical Trial Results in Pediatrics with HIV-1
Trial GS-US-216-0128 was a Phase 2/3 multicenter, open-label trial to evaluate the pharmacokinetics, safety, and efficacy of darunavir co-administered with cobicistat in adolescents aged 12 years and older (Cohort 1) and children aged 6 to less than 12 years old (Cohort 2) with HIV-1 who were virologically suppressed and had a baseline estimated creatinine clearance ≥90 mL/min/1.73 m 2. Participants were on a stable antiretroviral regimen including at least 2 NRTIs (for at least 3 months).
Cohort 1:The median age of participants was 14 years (range 12–16 years), median weight was 60 kg (range 45–78 kg), and 43% were male. At baseline, all had plasma HIV-1 RNA <50 copies/mL. At Week 48, 86% (6/7) of participants remained suppressed (HIV-1 RNA <50 copies/mL), and 1 had missing data. From a median baseline CD4+ cell count and CD4+% of 1,117 cells/mm 3(range 658 to 2,416 cells/mm 3) and 45% (range 28% to 56%), respectively, the median change from baseline in CD4+ cell count and CD4+% at Week 48 was -342 cells/mm 3(range -1,389 to 219 cells/mm 3) and -6% (range -12% to 5%), respectively. All 6 participants with available data had CD4+ cell counts above 800 cells/mm 3at Week 48.
Cohort 2:The median age of participants was 10 years (range 7–11 years), median weight was 31.7 kg (range 29.1–45.4 kg), and 75% were female. At baseline, all had plasma HIV-1 RNA <50 copies/mL. At Week 48, all participants remained suppressed (HIV-1 RNA <50 copies/mL). From a median baseline CD4+ cell count and CD4+% of 998 cells/mm 3(range 439 to 1113 cells/mm 3) and 40% (range 29.1% to 48.5%), respectively, the median change from baseline in CD4+ cell count and CD4+% at Week 48 was -20 cells/mm 3(range -318 to 245) and 0.75% (-11.40 to 3.20), respectively.
-
16 HOW SUPPLIED/STORAGE AND HANDLING
PREZCOBIX ®(darunavir and cobicistat) tablets, 800/150 mg, are supplied as pink, oval-shaped, film-coated tablets debossed with "800" on one side and "TG" on the other side. Bottle of 30 tablets (NDC 59676-575-30).
PREZCOBIX ®(darunavir and cobicistat) tablets, 675/150 mg, are supplied as green to dark green, oval-shaped, scored film-coated tablet debossed with “675” on one side and “TG” on the other side. Bottle of 30 tablets (NDC 59676-578-30).
-
17 PATIENT COUNSELING INFORMATION
Advise the patient to read the FDA-approved patient labeling (Patient Information).
Instructions for Use
Advise patients to take PREZCOBIX with food every day on a regular dosing schedule, as missed doses can result in development of resistance. Inform patients not to alter the dose of PREZCOBIX or discontinue therapy with PREZCOBIX without consulting their physician [see Dosage and Administration (2.2)].
Hepatotoxicity
Inform patients that drug-induced hepatitis (e.g., acute hepatitis, cytolytic hepatitis) and liver injury, including some fatalities, could potentially occur with PREZCOBIX. Advise patients to contact their healthcare provider immediately if signs and symptoms of liver problems develop [see Warnings and Precautions (5.1)] .
Severe Skin Reactions
Inform patients that skin reactions ranging from mild to severe, including Stevens-Johnson Syndrome, drug rash with eosinophilia and systemic symptoms, and toxic epidermal necrolysis, could potentially occur with PREZCOBIX. Advise patients to contact their healthcare provider immediately if signs or symptoms of severe skin reactions develop, including but not limited to severe rash or rash accompanied with fever, general malaise, fatigue, muscle or joint aches, blisters, oral lesions, and/or conjunctivitis [see Warnings and Precautions (5.2)] .
Renal Impairment
Inform patients that renal impairment, including cases of acute renal failure and Fanconi syndrome, has been reported when cobicistat is used in combination with a tenofovir DF-containing regimen [see Warnings and Precautions (5.4)] .
Pregnancy
Advise patients that PREZCOBIX is not recommended during pregnancy and to alert their healthcare provider if they get pregnant while taking PREZCOBIX [see Use in Specific Populations (8.1)] . Inform patients that there is an antiretroviral pregnancy registry to monitor fetal outcomes of pregnant individuals exposed to PREZCOBIX [see Use in Specific Populations (8.1)].
Lactation
Inform patients that the potential risks of breastfeeding include: (1) HIV-1 transmission (in infants without HIV-1), (2) developing viral resistance (in infants with HIV-1), and (3) serious adverse reactions in a breastfed infant similar to those seen in adults [see Use in Specific Populations (8.2)] .
Drug Interactions
PREZCOBIX may interact with many drugs; therefore, inform patients of the potential serious drug interactions with PREZCOBIX, and that some drugs are contraindicated with PREZCOBIX and other drugs may require dosage adjustment. Advise patients to report to their healthcare provider the use of any other prescription or nonprescription medication or herbal products, including St. John's wort.
Immune Reconstitution Syndrome
Advise patients to inform their healthcare provider immediately of any symptoms of infection, as in some patients with advanced HIV infection (AIDS), signs and symptoms of inflammation from previous infections may occur soon after anti-HIV treatment is started [see Warnings and Precautions (5.10)] .
Fat Redistribution
Inform patients that redistribution or accumulation of body fat may occur in patients receiving antiretroviral therapy, including PREZCOBIX and that the cause and long-term health effects of these conditions are not known at this time [see Warnings and Precautions (5.9)] .
- SPL UNCLASSIFIED SECTION
-
PATIENT PACKAGE INSERT
This Patient Information has been approved by the U.S. Food and Drug Administration. Revised: 03/2025 PATIENT INFORMATION
PREZCOBIX ®(prez-koe-bix)
(darunavir and cobicistat)
tabletsWhat is the most important information I should know about PREZCOBIX?
PREZCOBIX may cause serious side effects, including:- Liver problems. People taking PREZCOBIX may develop liver problems which may be life-threatening and can lead to death. Your healthcare provider should do blood tests before and during your treatment with PREZCOBIX. If you have chronic hepatitis B or C infection or other liver problems, your healthcare provider may check your blood tests more often because you have an increased risk of developing liver problems. Tell your healthcare provider right away if you develop any new or worsening signs or symptoms of liver problems, including:
- tiredness
- dark (tea colored) urine
- yellowing of your skin or whites of your eyes
- pale colored stools (bowel movements)
- nausea
- vomiting
- pain or tenderness on your right side below your ribs
- loss of appetite
- Severe skin reactions or rash.Skin rash is common during treatment with PREZCOBIX but can become severe and require treatment in a hospital. Call your healthcare provider right away if you develop a rash. Stop taking PREZCOBIX and call your healthcare provider right away if you develop a severe rash or a rash with any of the following symptoms:
- fever
- tiredness
- muscle or joint pain
- blisters or skin lesions
- mouth sores or ulcers
- red or inflamed eyes, like "pink eye" (conjunctivitis)
- Kidney problems. PREZCOBIX when taken with certain other medicines can cause new or worsening kidney problems, including kidney failure. Your healthcare provider should check your kidneys before you start and during treatment with PREZCOBIX.
What is PREZCOBIX?
PREZCOBIX is a prescription medicine that is used with other human immunodeficiency virus (HIV-1) medicines to treat HIV-1 in adults and in children who weigh at least 55 pounds (25 kg).
HIV-1 is the virus that causes Acquired Immune Deficiency Syndrome (AIDS).
PREZCOBIX contains the prescription medicines darunavir and cobicistat.
It is not known if PREZCOBIX is safe and effective in children weighing less than 55 pounds (25 kg).Do not take PREZCOBIX with any medicine that contains: - alfuzosin
- carbamazepine
- colchicine, if you have liver or kidney problems
- dronedarone
- elbasvir and grazoprevir
- ergot-containing medicines:
- dihydroergotamine
- ergotamine tartrate
- methylergonovine
- ivabradine
- lomitapide
- lovastatin
- lurasidone
- midazolam, when taken by mouth
- naloxegol
- phenobarbital
- phenytoin
- pimozide
- ranolazine
- rifampin
- sildenafil, when used for the treatment of pulmonary arterial hypertension (PAH)
- simvastatin
- St. John's wort ( Hypericum perforatum)
- triazolam
Before taking PREZCOBIX, tell your healthcare provider about all your medical conditions, including if you: - have liver problems, including hepatitis B or hepatitis C, or liver disease (cirrhosis)
- have kidney problems
- are allergic to sulfa (sulfonamide)
- have diabetes
- have hemophilia
- are pregnant or plan to become pregnant.
- It is not known if PREZCOBIX will harm your unborn baby.
- PREZCOBIX should not be used during pregnancy because the PREZCOBIX levels in your blood may be lower during pregnancy and may not control your HIV-1.
- Tell your healthcare provider right away if you become pregnant during treatment with PREZCOBIX.
- Your healthcare provider will prescribe different medicines if you become pregnant during treatment with PREZCOBIX.
- Hormonal forms of birth control, such as injections, vaginal rings or implants, contraceptive patches, and some birth control pills may not work during treatment with PREZCOBIX. Talk to your healthcare provider about forms of birth control that may be used during treatment with PREZCOBIX.
- Pregnancy Exposure Registry: There is a pregnancy exposure registry for people who take PREZCOBIX during pregnancy. The purpose of the registry is to collect information about the health of you and your baby. Talk to your healthcare provider about how you can take part in this registry.
- are breastfeeding or plan to breastfeed. It is not known if PREZCOBIX passes into your breast milk.
- Talk to your healthcare provider about the following risks of breastfeeding during treatment with PREZCOBIX:
- The HIV-1 virus may pass to your baby if your baby does not have HIV-1.
- The HIV-1 virus may become harder to treat if your baby has HIV-1.
- Your baby may get side effects from PREZCOBIX.
- Talk to your healthcare provider about the following risks of breastfeeding during treatment with PREZCOBIX:
- You can ask your healthcare provider or pharmacist for a list of medicines that interact with PREZCOBIX.
- Do not start taking a new medicine without telling your healthcare provider. Your healthcare provider can tell you if it is safe to take PREZCOBIX with other medicines.
How should I take PREZCOBIX? - Take PREZCOBIX exactly as your healthcare provider tells you to take it.
- Do not change your dose or stop taking PREZCOBIX without talking to your healthcare provider. Stay under a healthcare provider’s care during treatment with PREZCOBIX.
- Take PREZCOBIX 1 time a day with food.
- For children weighing at least 55 pounds (25 kg) who cannot swallow a tablet whole:The scored tablet can be split by hand into 2 pieces. The score line will help you break the tablet. After splitting the tablet, swallow the 2 pieces right away to get the full dose of PREZCOBIX.
- When your PREZCOBIX supply starts to run low, get more from your healthcare provider or pharmacy. This is very important because the amount of virus in your blood may increase if the medicine is stopped for even a short time. The virus may develop resistance to PREZCOBIX and become harder to treat.
- If you take too much PREZCOBIX, call your healthcare provider or go to the nearest hospital emergency room right away.
What are the possible side effects of PREZCOBIX? PREZCOBIX may cause serious side effects, including: - Diabetes and high blood sugar (hyperglycemia).New or worsening diabetes and hyperglycemia have happened in people who take protease inhibitors including PREZCOBIX. Some people have had to start taking medicine to treat diabetes or have had to change their diabetes medicine. Tell your healthcare provider if you notice an increase in thirst or if you start urinating more often during treatment with PREZCOBIX.
- Changes in body fat can happen in people who take HIV-1 medications. The changes may include an increased amount of fat in the upper back and neck ("buffalo hump"), breast, and around the middle of your body (trunk). Loss of fat from the legs, arms, and face may also happen. The exact cause and long-term health effects of these conditions are not known.
- Changes in your immune system (Immune Reconstitution Syndrome) can happen when you start taking HIV-1 medicines. Your immune system may get stronger and begin to fight infections that have been hidden in your body for a long time. Tell your healthcare provider right away if you have any symptoms of infection after starting your HIV-1 medicine.
- Increased bleeding for people with hemophilia.Some people with hemophilia have increased bleeding with protease inhibitors including PREZCOBIX.
- diarrhea
- nausea
- rash
- headache
- stomach-area (abdominal) pain
- vomiting
These are not all of the possible side effects of PREZCOBIX.
Call your doctor for medical advice about side effects. You may report side effects to FDA at 1-800-FDA-1088.How should I store PREZCOBIX? - Store PREZCOBIX tablets at room temperature between 68 °F to 77 °F (20 °C to 25 °C).
General information about the safe and effective use of PREZCOBIX.
Medicines are sometimes prescribed for purposes other than those listed in a Patient Information leaflet. Do not use PREZCOBIX for a condition for which it was not prescribed. Do not give PREZCOBIX to other people, even if they have the same symptoms that you have. It may harm them.
You can ask your pharmacist or healthcare provider for information about PREZCOBIX that is written for health professionals.What are the ingredients in PREZCOBIX?
Active ingredients: darunavir and cobicistat
Inactive ingredients 800 mg/150 mg tablet:colloidal silicon dioxide, crospovidone, hypromellose, magnesium stearate, and silicified microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide red, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide.
Inactive ingredients 675 mg/150 mg tablet:colloidal silicon dioxide, croscarmellose sodium, magnesium stearate, and microcrystalline cellulose. The tablets are film-coated with a coating material containing iron oxide black, iron oxide yellow, polyethylene glycol, polyvinyl alcohol (partially hydrolyzed), talc, and titanium dioxide.
Manufactured for: Janssen Products, LP, Horsham, PA 19044, USA
For patent information: www.janssenpatents.com © Johnson & Johnson and its affiliates 2015, 2025
For more information call 1-800-526-7736. - PRINCIPAL DISPLAY PANEL - 800 mg/150 mg Tablet Bottle Label
- PRINCIPAL DISPLAY PANEL - 675 mg/150 mg Tablet Bottle Carton
-
INGREDIENTS AND APPEARANCE
PREZCOBIX
darunavir ethanolate and cobicistat tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:59676-575 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DARUNAVIR ETHANOLATE (UNII: 33O78XF0BW) (DARUNAVIR - UNII:YO603Y8113) DARUNAVIR 800 mg COBICISTAT (UNII: LW2E03M5PG) (COBICISTAT - UNII:LW2E03M5PG) COBICISTAT 150 mg Inactive Ingredients Ingredient Name Strength HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSPOVIDONE (120 .MU.M) (UNII: 68401960MK) MAGNESIUM STEARATE (UNII: 70097M6I30) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) TALC (UNII: 7SEV7J4R1U) FERRIC OXIDE RED (UNII: 1K09F3G675) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Product Characteristics Color pink Score no score Shape OVAL Size 23mm Flavor Imprint Code 800;TG Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59676-575-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 01/31/2015 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA205395 01/31/2015 PREZCOBIX
darunavir ethanolate and cobicistat tablet, film coatedProduct Information Product Type HUMAN PRESCRIPTION DRUG Item Code (Source) NDC:59676-578 Route of Administration ORAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DARUNAVIR ETHANOLATE (UNII: 33O78XF0BW) (DARUNAVIR - UNII:YO603Y8113) DARUNAVIR 675 mg COBICISTAT (UNII: LW2E03M5PG) (COBICISTAT - UNII:LW2E03M5PG) COBICISTAT 150 mg Inactive Ingredients Ingredient Name Strength HYPROMELLOSE, UNSPECIFIED (UNII: 3NXW29V3WO) MICROCRYSTALLINE CELLULOSE (UNII: OP1R32D61U) SILICON DIOXIDE (UNII: ETJ7Z6XBU4) CROSCARMELLOSE SODIUM (UNII: M28OL1HH48) MAGNESIUM STEARATE (UNII: 70097M6I30) POLYVINYL ALCOHOL, UNSPECIFIED (UNII: 532B59J990) TITANIUM DIOXIDE (UNII: 15FIX9V2JP) POLYETHYLENE GLYCOL, UNSPECIFIED (UNII: 3WJQ0SDW1A) TALC (UNII: 7SEV7J4R1U) FERRIC OXIDE YELLOW (UNII: EX438O2MRT) FERROSOFERRIC OXIDE (UNII: XM0M87F357) Product Characteristics Color green Score no score Shape OVAL Size 21mm Flavor Imprint Code 675;TG Contains Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:59676-578-30 30 in 1 BOTTLE; Type 0: Not a Combination Product 03/21/2025 Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date NDA NDA205395 03/21/2025 Labeler - Janssen Products LP (804684207) Establishment Name Address ID/FEI Business Operations Gilead Alberta ULC 207452996 api manufacture(59676-575) Establishment Name Address ID/FEI Business Operations Cilag AG 483237103 api manufacture(59676-575) Establishment Name Address ID/FEI Business Operations Yuhan Chemical Inc. 687831958 api manufacture(59676-575, 59676-578) Establishment Name Address ID/FEI Business Operations Janssen Ortho LLC 805887986 manufacture(59676-575) , analysis(59676-575) Establishment Name Address ID/FEI Business Operations Janssen Pharmaceutical Sciences Unlimited Company 985639841 api manufacture(59676-575) Establishment Name Address ID/FEI Business Operations Porton Pharma Solutions Ltd 528192390 api manufacture(59676-575, 59676-578) Establishment Name Address ID/FEI Business Operations Thermo Fisher Scientific Cork Limited 985745028 api manufacture(59676-575, 59676-578) Establishment Name Address ID/FEI Business Operations Patheon, Inc. 240769596 manufacture(59676-578) , pack(59676-578)