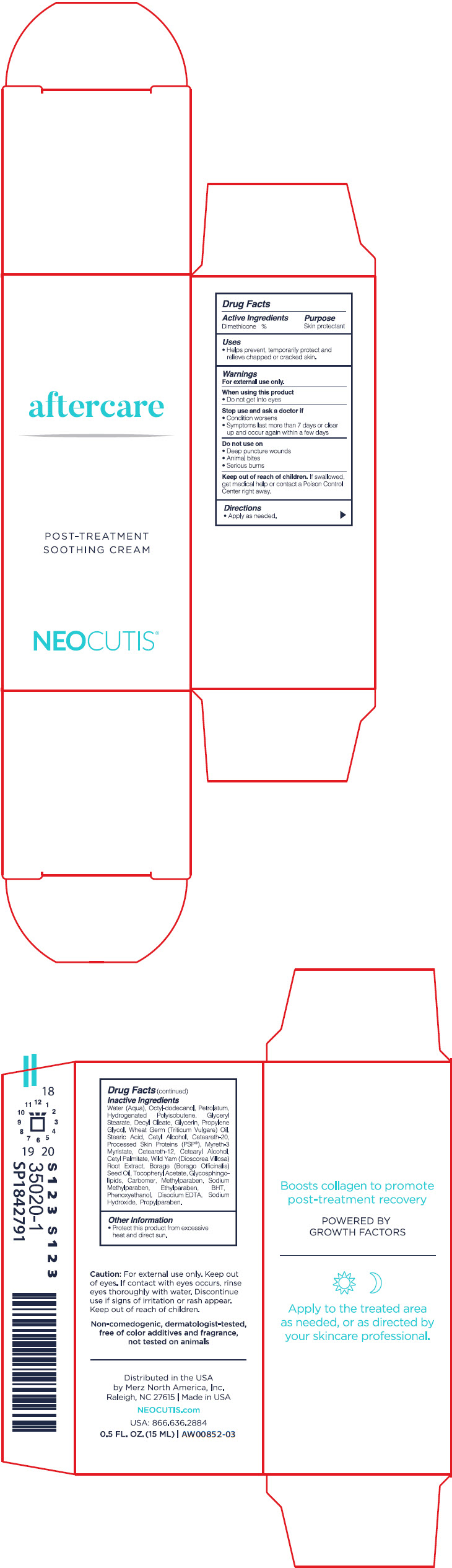
Label: AFTER CARE- dimethicone cream
- NDC Code(s): 46783-004-04, 46783-004-15, 46783-004-20, 46783-004-30
- Packager: Merz North America
- Category: HUMAN OTC DRUG LABEL
- DEA Schedule: None
- Marketing Status: OTC monograph final
DISCLAIMER: Most OTC drugs are not reviewed and approved by FDA, however they may be marketed if they comply with applicable regulations and policies. FDA has not evaluated whether this product complies.
Drug Label Information
Updated September 27, 2023
If you are a consumer or patient please visit this version.
- Download DRUG LABEL INFO: PDF XML
- Official Label (Printer Friendly)
- SPL UNCLASSIFIED SECTION
- Active ingredients
- Purpose
- Uses
- Warnings
- Directions
-
Inactive ingredients
Water (Aqua), Octyl-Dodecanol, Petrolatum, Hydrogenated Polyisobutene, Glyceryl Stearate, Decyl Oleate, Glycerin, Propylene Glycol, Wheat Germ (Triticum Vulgare) Oil, Stearic Acid, Cetyl Alcohol, Ceteareth-20, Processed Skin Proteins (PSP(R)), Myreth-3 Myristate, Ceteareth-12, Cetearyl Alcohol, Cetyl Palmitate, Wild Yam (Dioscorea Villosa) Root Extract, Borage (Borago Officinalis) Seed Oil, Tocopheryl Acetate, Glycosphingolipids, Carbomer, Methylparaben, Sodium Methylparaben, Ethylparaben, BHT, Phenoxyethanol, Disodium EDTA, Sodium Hydroxide, Propylparaben.
- Other information
- SPL UNCLASSIFIED SECTION
- PRINCIPAL DISPLAY PANEL - 15 ML Bottle Carton
-
INGREDIENTS AND APPEARANCE
AFTER CARE
dimethicone creamProduct Information Product Type HUMAN OTC DRUG Item Code (Source) NDC:46783-004 Route of Administration TOPICAL Active Ingredient/Active Moiety Ingredient Name Basis of Strength Strength DIMETHICONE (UNII: 92RU3N3Y1O) (DIMETHICONE - UNII:92RU3N3Y1O) DIMETHICONE 4 mg in 1 mL Inactive Ingredients Ingredient Name Strength WATER (UNII: 059QF0KO0R) OCTYLDODECANOL (UNII: 461N1O614Y) PETROLATUM (UNII: 4T6H12BN9U) HYDROGENATED POLYBUTENE (1300 MW) (UNII: 7D1YQ9Y5EZ) GLYCERYL MONOSTEARATE (UNII: 230OU9XXE4) DECYL OLEATE (UNII: ZGR06DO97T) GLYCERIN (UNII: PDC6A3C0OX) PROPYLENE GLYCOL (UNII: 6DC9Q167V3) WHEAT GERM OIL (UNII: 14C97E680P) STEARIC ACID (UNII: 4ELV7Z65AP) CETYL ALCOHOL (UNII: 936JST6JCN) POLYOXYL 20 CETOSTEARYL ETHER (UNII: YRC528SWUY) HUMAN SKIN PROTEINS, PARTIALLY HYDROLYZED (UNII: G9813R29TW) MYRETH-3 MYRISTATE (UNII: O2C2MN32O6) CETEARETH-12 (UNII: 7V4MR24V5P) CETOSTEARYL ALCOHOL (UNII: 2DMT128M1S) CETYL PALMITATE (UNII: 5ZA2S6B08X) DIOSCOREA VILLOSA TUBER (UNII: IWY3IWX2G8) BORAGE SEED OIL (UNII: F8XAG1755S) .ALPHA.-TOCOPHEROL ACETATE (UNII: 9E8X80D2L0) CARBOMER HOMOPOLYMER, UNSPECIFIED TYPE (UNII: 0A5MM307FC) METHYLPARABEN (UNII: A2I8C7HI9T) METHYLPARABEN SODIUM (UNII: CR6K9C2NHK) ETHYLPARABEN (UNII: 14255EXE39) BUTYLATED HYDROXYTOLUENE (UNII: 1P9D0Z171K) PHENOXYETHANOL (UNII: HIE492ZZ3T) EDETATE DISODIUM ANHYDROUS (UNII: 8NLQ36F6MM) SODIUM HYDROXIDE (UNII: 55X04QC32I) PROPYLPARABEN (UNII: Z8IX2SC1OH) Packaging # Item Code Package Description Marketing Start Date Marketing End Date 1 NDC:46783-004-15 1 in 1 CARTON 04/01/2019 1 15 mL in 1 BOTTLE; Type 0: Not a Combination Product 2 NDC:46783-004-20 1 in 1 CARTON 04/01/2019 2 200 mL in 1 BOTTLE; Type 0: Not a Combination Product 3 NDC:46783-004-04 4 mL in 1 TUBE; Type 0: Not a Combination Product 06/11/2019 4 NDC:46783-004-30 1 in 1 CARTON 10/02/2023 4 30 mL in 1 BOTTLE; Type 0: Not a Combination Product Marketing Information Marketing Category Application Number or Monograph Citation Marketing Start Date Marketing End Date OTC monograph final part347 04/01/2019 Labeler - Merz North America (028147846)